Ensuring safe vascular access for the delivery of intravenous (IV) therapies is a cornerstone of everyday neonatal practice.1,2 IV access is obtained via peripheral and central veins with peripheral vein cannulation being the most frequently performed procedure in the Neonatal Intensive Care Unit (NICU).3,4 However, obtaining and maintaining IV access in the neonatal patient population is often challenging and can be a common cause of iatrogenic complication and injury.5
While many complications are minor, the risk of peripheral IV infiltration and extravasation (PIVIE) injury has the potential to cause more serious harm. The reported risk of a peripheral IV-related complication in this patient population is variable with some authors of single and multicenter studies reporting complication rates of 63.5% to 75%.3,5–6 Pooled figures for extravasation incidence from meta-analyses also show considerable heterogeneity.7 In part, this variation is due to differing classifications of what constitutes a complication, definitional overlap, and amalgamation of complication incidence in reported figures (eg infiltration, extravasation, and leakage reported together). Furthermore, various confounding factors such as weight, gestational age, the nature of the infusion fluid, the use of vascular access technology to evaluate potential insertion sites, chosen vein, underlying pathology, and practitioner skill all influence risk.2,8 According to several sources, PIVIE is implicated in 65% of neonatal IV complications.3,9,10,11 Because of this high prevalence, numerous evidence-based measures have been implemented to reduce the likelihood and severity of complications. These interventions include care bundles, site selection, and evaluation tools (eg VeinViewer© [Christies], and the touch, look, compare mnemonic), catheter design and material innovations, dynamic inline occlusion pressure monitoring, and insertion site optical monitoring technology (eg ivWatch®). In the neonatal literature, treatment of PIVIE is underreported.12 In the case we describe, we discuss our current practice in the treatment of PIVIE.
The case
A term male patient was born by emergency cesarean section to a 28-year-old primigravida mother for decreased fetal movement. The infant was born in good condition and did not require resuscitation. Vital signs were within normal expectations. Apgar scores were 9 and 10 at 1 and 5 minutes of life, respectively. His birthweight was 2299 g (fifth percentile), and his head circumference was 32 cm (20th percentile). Physical examination indicated asymmetric intrauterine growth restriction but nothing else of note. In accord with institutional policy to ensure mother and baby remain together whenever possible, he was risk assessed using local guidelines and admitted to the postnatal areas under the direct 24-hour supervision of the resident medical and nursing staff. Despite measures taken to prevent low blood sugar, he was later admitted to the NICU for further investigation and treatment of hypoglycemia. As per local practice, a peripheral IV was inserted and an infusion of dextrose (10%) commenced.
On the third day of life, the IV cannula was resited in the left cephalic vein using a 26-gauge polyurethane catheter. Dextrose 10% water infusing at a rate of 9.7 mL/h was recommenced. As is our usual practice, the infusion flow was controlled by an infusion pump using dynamic inline pressure monitoring and the cannula insertion site itself monitored with an ivWatch sensor.
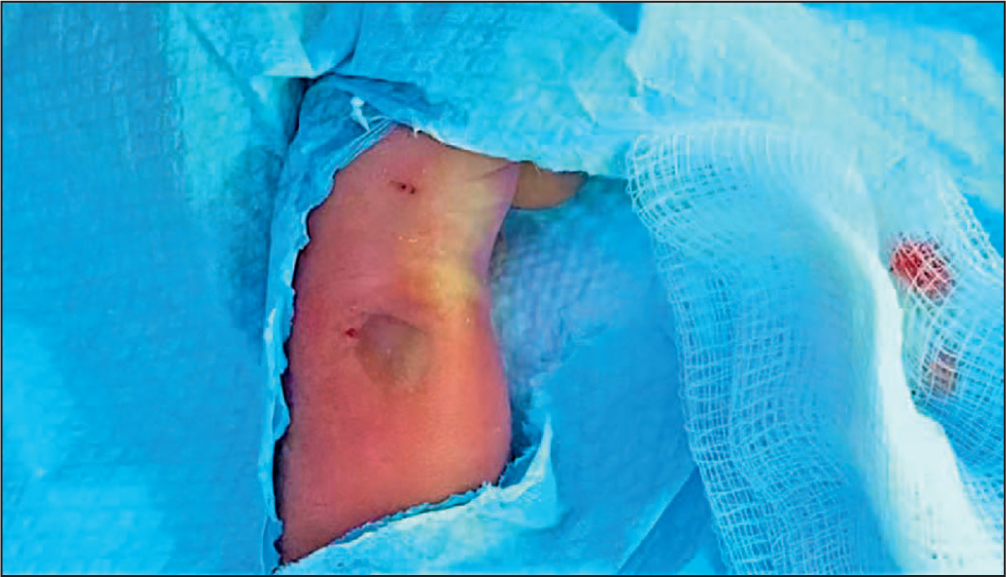
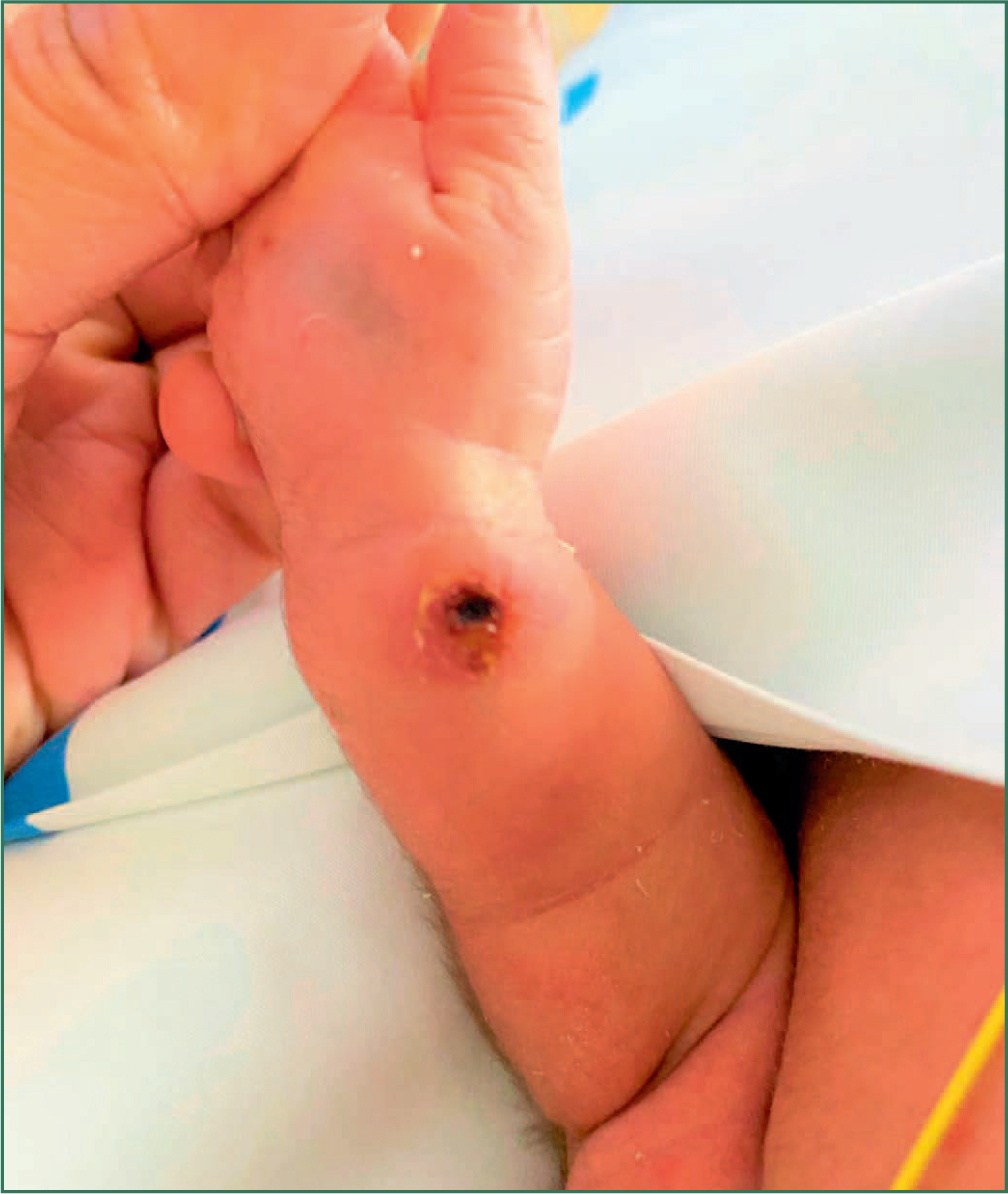
Three hours after recommencing the infusion, swelling with erythema, bruising, and blanching was observed at the insertion site (Figure 1). The cannula was removed and a decision made on further treatment. Parents of the infant were briefed about the incident and its treatment, and consent was obtained for photography.
Diagnosis and management
IV extravasation is defined as occurring after unintentional administration of a vesicant fluid into surrounding vascular tissue.13 With any PIVIE injury, it is important to assess the extent and severity of the injury to inform treatment decisions. Several descriptive and more objective staging techniques are reported in the literature.14,15 Our preferred approach is based upon the Cincinnati Children's Medical Center tool.14 Using this tool, the arm length (regardless of the site of infiltration/extravasation) is measured from axilla to the tip of the longest finger (value of Y). Secondly, the volume of the infiltrated/extravasated fluid is assessed by defining the edges of swelling by palpation/visual inspection, measuring of the longest dimension (value of X), and lastly, dividing X by Y and multiplying the outcome by 100 to get a percentage which is used in a treatment algorithm.
Treatment depends on the severity of the PIVIE and on the characteristics of the extravasated fluids used. In the described case, the PIVIE score was 30% (Figure 1), and treatment with hyaluronidase to aid dispersion of the hyperosmolar extravasated fluid was indicated.1 Hyaluronidase, which is an enzyme involved in the catabolism of hyaluronic acid (a key component of the extracellular matrix), has wide-ranging physiological roles in the body from reproduction to aging, and is also implicated in pathology processes.16 The enzyme has commercial uses in laboratories, the cosmetics industry, in plastic surgery, and therapeutically.16,17,18,19 Clinically, it is used to increase the bioavailability of some drugs or in the treatment of extravasation; it does this by facilitating swifter dispersion and absorption of medications or extravasated fluids through degradation of the extracellular matrix.1,17,19
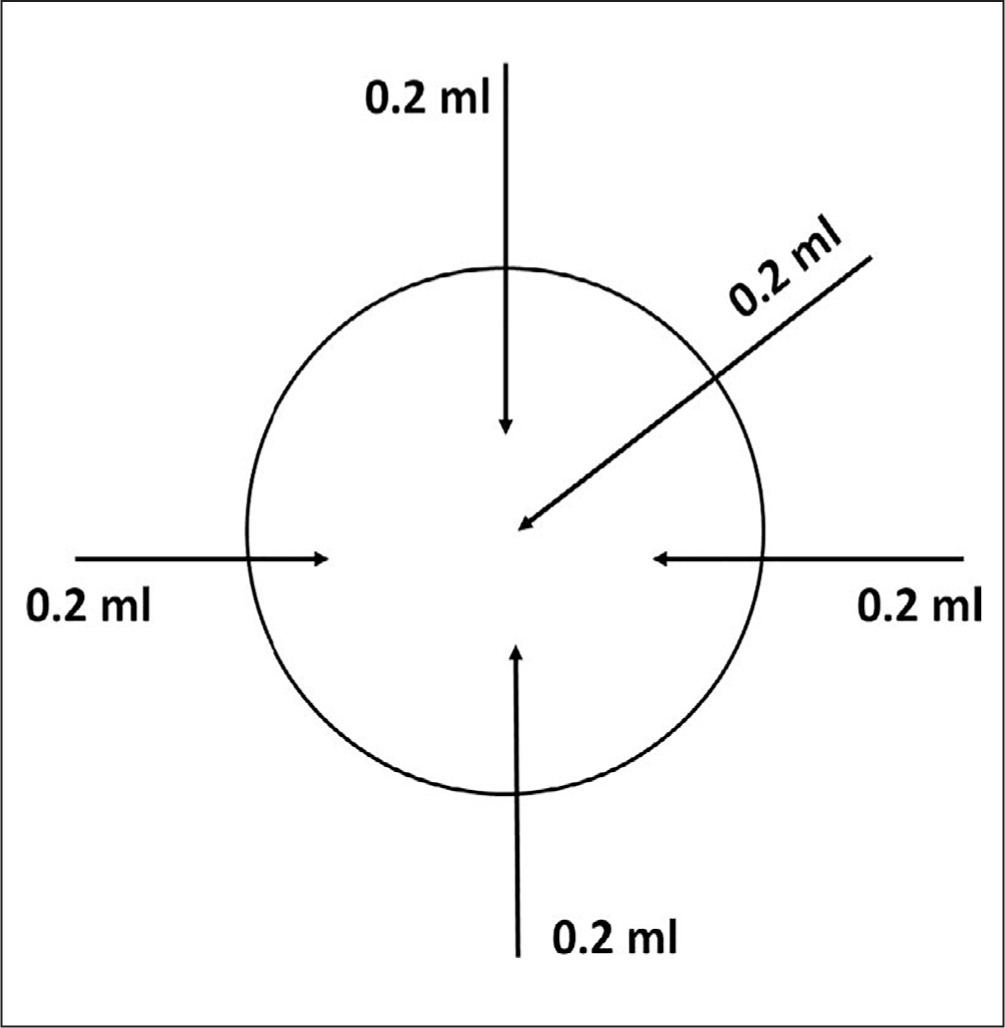
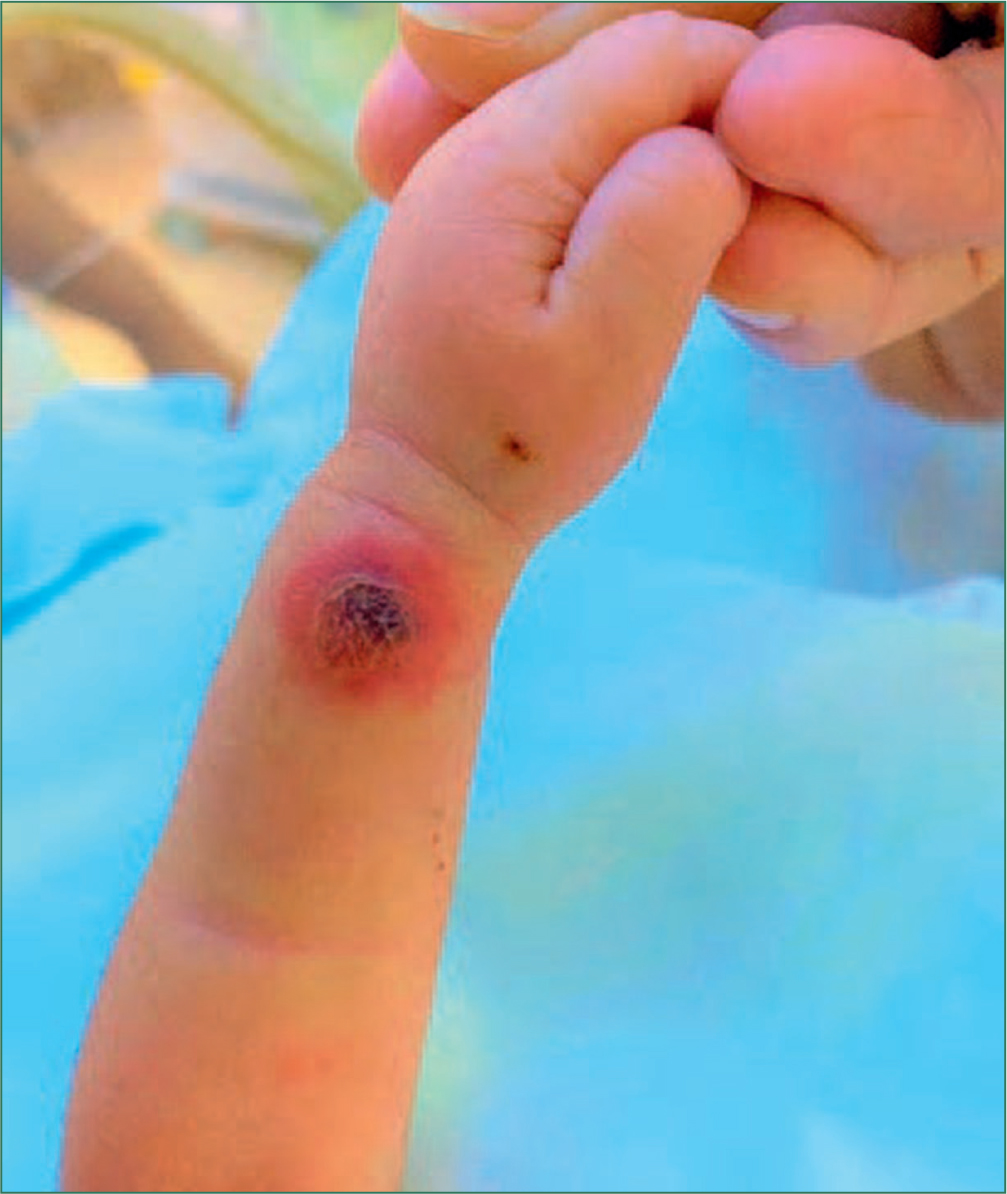
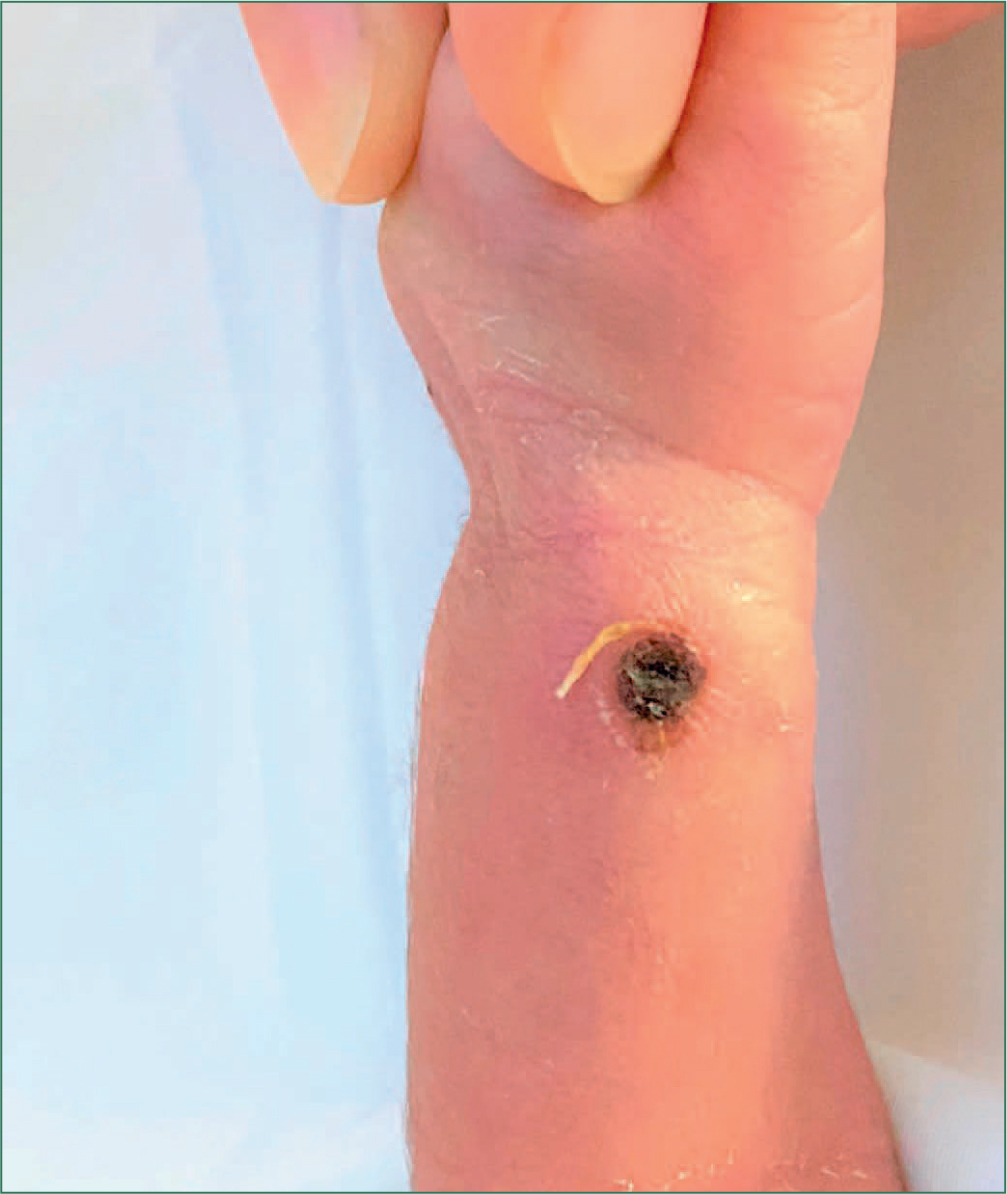
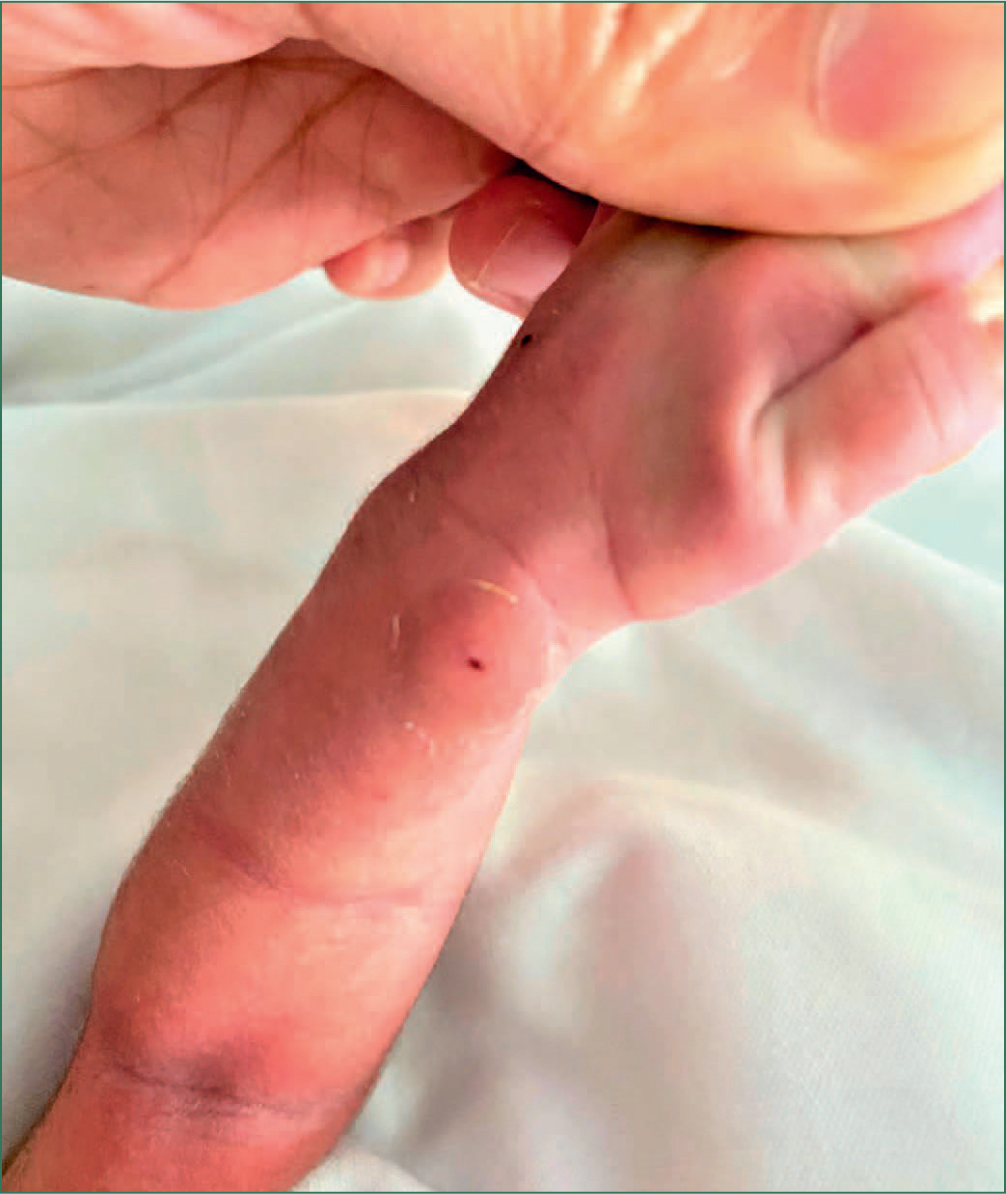
Hyaluronidase was prepared according to local protocol (based on international recommendations) by diluting with 1 mL of sodium chloride 0.9%.1 The hyaluronidase was then injected in rotation (Figure 2) subcutaneously around and through the area of the injury site; the needle was changed before each injection. Subsequent images (Figures 3–7) illustrate wound healing progression over a period of 2 weeks.
Discussion
Frequent and repeated invasive peripherally intravenous catheter (PIVC) insertions cause discomfort and increase infant pain experience with increased risk of complications like infection due to repeated breakages of the skin.6 Commonly used alternative vascular access devices in neonates include extended peripheral IV catheters and peripherally inserted central catheters. Both are considered safe, and success rates of insertions are relatively high with low complication rates.4,20 A goal of vascular access is to insert the Right vascular access device for the right therapy/fluids, for the right duration, and for the right patient. In our case, the IV fluid was a hypertonic acidic concentration of dextrose,1,21 which increased the risk of extravasation harm. With hindsight and given the duration of IV therapy for this infant and the characteristics of infused fluids, it might have been more optimal to have selected an alternative vascular access device.
Interestingly, in this case, the ivWatch did not issue an infiltration alert before observation by nursing staff; it is not clear why this happened. One explanation might be that the sensor was incorrectly positioned. However, on investigation, this does not seem likely. A pilot study22 in our unit evaluating the use of the ivWatch sensor technology in a neonatal setting reported a sensitivity of 93.3% for detecting infiltration, which, despite this event, is within operational expectations for the equipment and underlines the need for continued observational vigilance of IV infusion sites. Nonetheless, this situation leaves the possibility for further refinement of the algorithm and reinforces messages about the continual need for vigilant observation at the bedside to complement technological aids.
Consensus about the optimal treatment of neonatal extravasation is limited, in part due to a lack of robust evidence to support practice and inconsistent wound evaluation.12 Current treatment options consist of a range of interventions including conservative (observation, affected limb elevation) saline washouts, injection of hyaluronidase (with or without washout), dressing, debridement, and plastic surgery. Routine investigation of this adverse incident highlighted a lack of consensus and debate around the choice of venous access device in this case. There was also some misinformation among practitioners about the merits and limitations of technical aids (inline pressure and optical site monitoring), revealing a tendency among some to overrely on these technological aids to detect infusion-related problems. Targeted bespoke educational updates using clinical, journal club, and reflective teaching and learning activities were implemented to address these issues.
Conclusions
The case reported here of an infant with a peripheral IV complication highlights ongoing debate about the best choice of vascular access device and the treatment of PIVIE injury. Nevertheless, this experience has raised awareness of the need for practitioners to be cognizant of the ‘5Rs for vascular access’, ie the Right device, for the Right vein, with the Right therapy, for the Right duration, for the Right patient. In retrospect, this replacement PIVC might not have been the optimal choice for this infant; an alternate device may have avoided this PIVIE event. Furthermore, this case emphasizes the need for continued vigilance of IV infusions and the avoidance of inadvertently overrelying on technology to detect issues. With early recognition, prompt intervention and management of the risk of long-term harm can be avoided.