Over 100 million people develop scars in the developed world alone each year because of 55 million elective and 25 million trauma operations (Gauglitz et al, 2011). Wound healing is a complex process that is described as taking place over three main phases: inflammation, proliferation and remodelling. According to Trace et al (2016), it is during the remodelling phase that collagen deposition occurs in an organised fashion that results in a normal scar.
Abnormal scarring occurs when there is a disruption in this process that can lead to excess collagen synthesis and deposition that results in a hypertrophic scar (De Decker et al, 2022). During the formation of scars, the epidermal layers of the skin will produce high levels of moisture in an attempt to hydrate the scar site. However, most of this moisture evaporates once it reaches the stratum corneum, or upper layer of the skin; this is due to damage to the stratum corneum (De Decker et al, 2022). This moisture loss triggers keratinocytes in the skin to produce collagen, leading to this disruption.
Scarring can cause cosmetic disfigurement, physical impairment, psychological distress and decreased quality of life (Finnerty et al, 2016; Trace et al, 2016; De Decker et al 2022). Sometimes even the smallest of scars may invoke a large psychological reaction from a patient and, conversely, patients with large injuries and more severe scarring, who may be expected to have more psychological difficulties, may cope more readily (Ngaage and Agius, 2018).
Hypertrophic scars are frequently seen as a result of prolonged spontaneous healing in deep dermal burns or after skin grafting in full-thickness burns (Cubison et al, 2006). Studies suggest that the incidence of hypertrophic scarring after these burns ranges from 32% to 72% (Finnerty et al, 2016; Pruksapong, 2020). This article will concentrate on hypertrophic scarring in minor burns, but the management options can be applied to any hypertrophic scar.
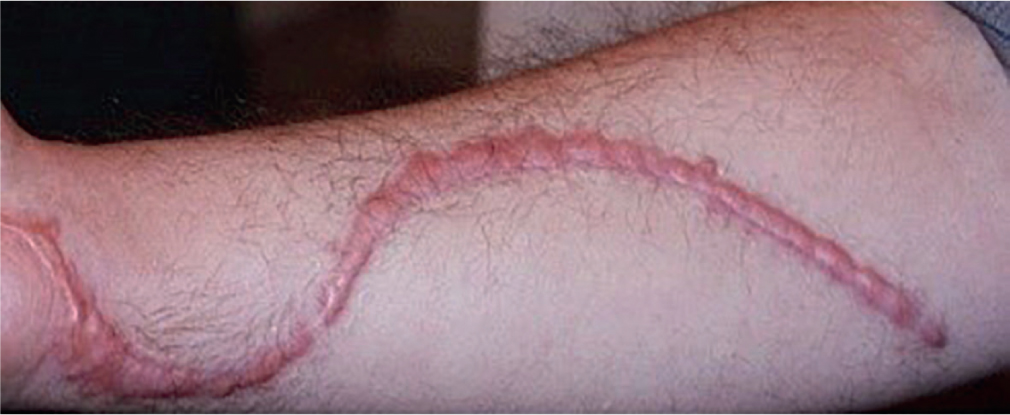
Hypertrophic scarring usually develops 4 to 6 weeks following wound infection, excess tension or traumatic skin injury (Figure 1). It has a period of rapid growth, and then it will regress over a couple of years. Infection can also play a key role in abnormal scar formation, because the presence of excessive bacterial growth in wounds has deleterious effects on the healing process (Panazyi et al, 2020). Bacteria in a wound present a metabolic burden (eg bioburden), as they compete with fibroblasts and macrophages for nutrients. If the bacterial level is too high, the wound can become chronic, and the healing process may fail, leading to the development of abnormal scarring (Panyani et al, 2020).
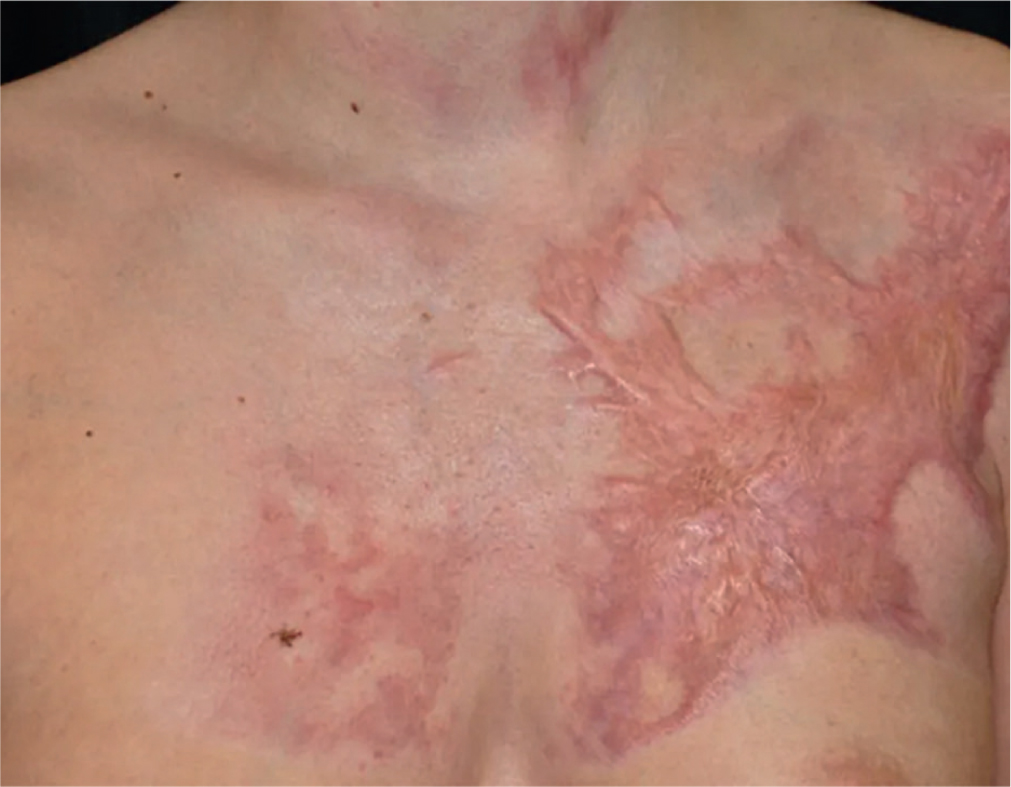
Hypertrophic scars occur over joints or in areas of high tension and are raised, red and rigid (Figure 2), often termed the three Rs (McGrory, 2013); other symptoms include altered pigmentation, scar contractures, altered sensation, pain and pruritus (itch). Hypertrophic scars stay within the margins of the original wound and reduce in size over time (Gauglitz et al, 2011).
Scar assessment
Since the 1950s, many scar assessment tools have been developed for burn scar assessment. The number of scar assessment tools available attests to the difficulty of finding one appropriate instrument. However, it is important that a scar is assessed so that efficacy of treatment can be determined (Table 1). Chae et al (2016) suggested that the ideal tool should be non-invasive, painless, easy-to-use and statistically reliable. However, there is still no consensus on the best method to achieve this goal for burn scarring. Very few tools have been validated for the paediatric population (Choo et al, 2021).
Table 1. Scar assessment tools
| Tool | Source |
|---|---|
| Vancouver Scar Scale | Sullivan et al (1990) |
| Modified Vancouver Scar Scale | Baryza and Baryza (1995) |
| Seattle Scale | Yeong et al (1997) |
| Manchester Scar Scale | Beausang et al (1998) |
| Smith Scale | Smith et al (1988) |
| Hamilton Scale | Crowe et al (1998) |
| Patient and Observer Scar Assessment Scale (POSAS) | Draaijers et al (2004) |
| Matching assessment of scars and photographs (MAPS) | Masters et al (2005) |
There are four key areas of scar assessment that all the tools have in common. These include vascularity, thickness, pliability and colour (Figure 3):
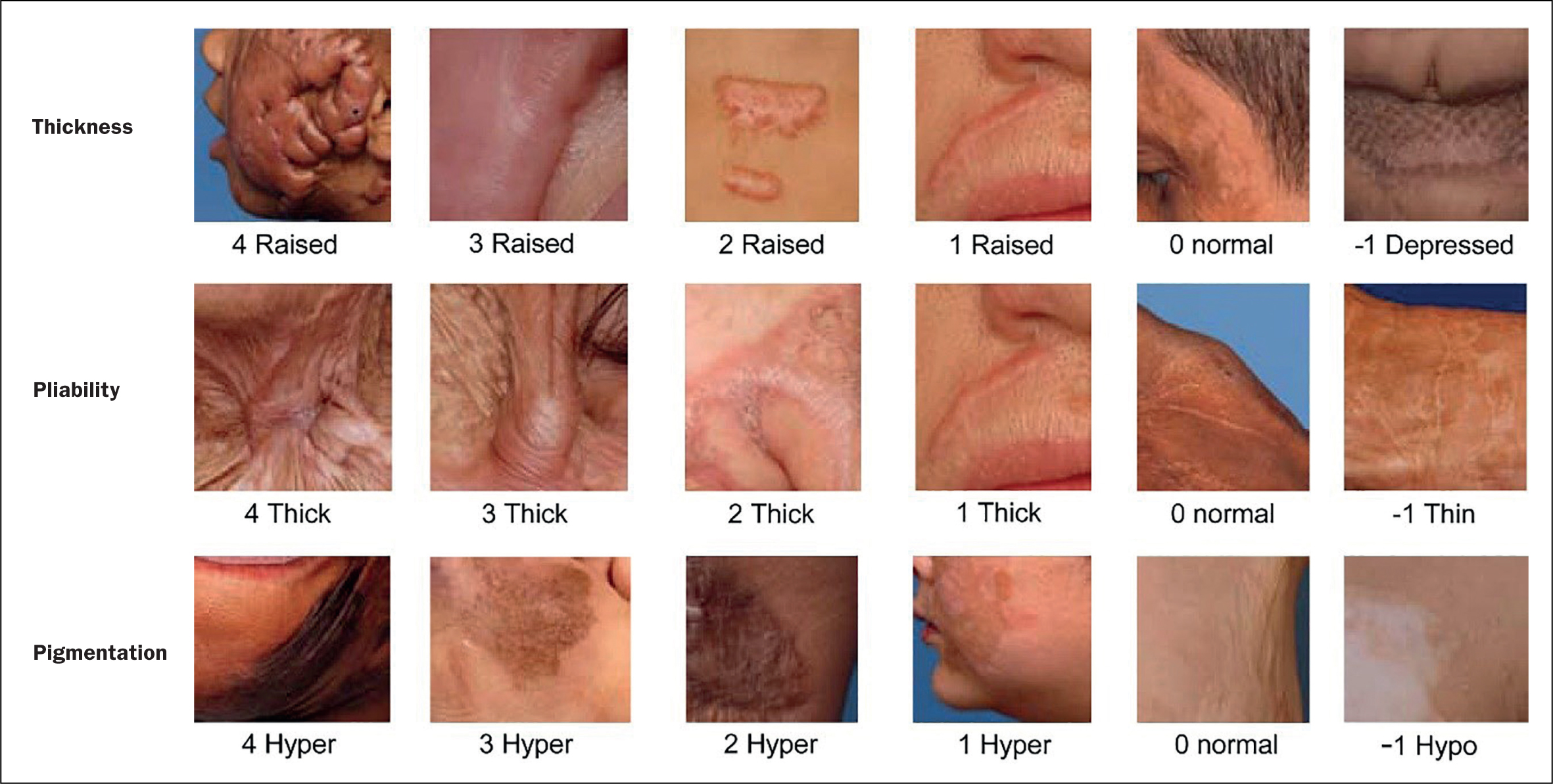
- Vascularity: the increased vascularisation of the scar (erythema) is a good indicator for scar activity in the early maturation phase (red). This can be assessed by using a capillary refill test by pressing on the scar and rating according to the return of the blood flow
- Colour (pigmentation): changes in their normal skin colour is a major concern of patients with scars; it causes significant body-image perception problems. This complication can cause low self-esteem and social isolation, especially in people with darker skin tones (Yenikomshian and Gibran, 2020). Pigmentation disorders are caused by variation in the concentration of melanocytes in the epidermal layer and their melanin production, but also by vascularity (Yamawaki, 2020). Significant pigmentation disorders may remain in the long term, such as hypo- or hyperpigmentation
- Thickness: the thickness of a scar is measured by comparing it with the adjacent normal skin in clinical settings, since scars are caused by the proliferation of collagen and fibroblasts in the dermis (Yamawaki, 2020)
- Pliability: scars commonly have a reduced pliability compared with normal skin. The increased stiffness of scars sometimes induces pain and itching because of the difference in their flexibility compared with the adjacent normal skin (Yamawaki, 2020). This may cause functional impairment, especially when scars are located on or around joints.
Scar management
The aims of scar management are to alleviate any symptoms, promote scar maturation, prevent any deformities or contractures, avoid growth retardation in children and reduce disfigurement (Teot et al 2020a). Hopefully, by achieving these aims it would facilitate psychological wellbeing and assist patients' reintegration into society. However, it is not always possible to achieve these aims and both the staff and the patient have to have realistic expectations, or this will adversely affect psychological wellbeing (Van Loey 2020).
There is a range of management options available to patients but many of these are in specialist burns services. This article will cover only those available outside specialist services.
Scar prevention
Prevention of scar occurrence should be the first line of scar management. Teot et al (2020a) suggested that prevention of abnormal scarring is more effective than treating it, which is often unsatisfying for the patient and adds an additional burden to healthcare expenses. Some seminal research undertaken by Deitch et al (1983) demonstrated that the time to healing of the burn was the most important factor in scar prevention, the degree of scarring being strongly dependent on the duration of healing. They demonstrated that there was a significantly enhanced risk of hypertrophic scarring if the burn was unhealed at 10 days, which increased to 80% for burns taking longer than 21 days to heal. This was supported by Cubison et al (2006), who found a strong correlation between time to healing and hypertrophic scarring in a study of over 500 paediatric patients with burns. Therefore, accurate depth assessment is essential to know which burns will heal within that timeframe and which burns may need surgical intervention to ensure healing within 3 weeks. Teot et al (2020a) suggested that deeper burns should be referred to a burn service and skin grafted as soon as possible.
The quality of immediate care has an important influence on the outcome of the burn wound in terms of scarring. A superficial injury can turn into a deeper burn due to poor general management. The choice of an appropriate dressing that prevents the burn wound drying out and prevents infection is essential. Infection has been closely linked with hypertrophic scar formation (Teot et al, 2020b), and, as such, an antimicrobial dressing is usually recommended for burn wounds. Burn wounds heal best in a moist but not wet environment that promotes epithelialisation and prevents cellular dehydration, which slows down healing. This can best be accomplished by applying either a topical agent or an occlusive dressing to minimise fluid loss (Yowler and Coffee, 2020).
Another important aspect is to promote function. Oedema is often a problem in burn patients, and regular exercise should be promoted to reduce this oedema (Voight et al, 2018). The use of slings for upper-extremity burns should not be advocated. Instead, the patient should be put at rest with their the arm or hand on pillows or cushions and sat with their legs elevated. The ability to promote range of movement (RoM) and undertake activities of daily living (ADL) is essential to maintain function (Kamolz and Jeschke, 2020).
Emollients
In hypertrophic scarring, there is damage to the dermal structures, such as sweat glands, hair follicles and sebaceous glands. This means that hypertrophic scars often fail to produce sweat or oils (Klotz et al, 2017). In addition, hypertrophic scars have a high trans-epidermal water loss (TEWL) when compared with normal skin, particularly in wounds that have healed by secondary intention (Anthonissen et al, 2013). The combination of these two factors means that hypertrophic scars appear dry, and it is for this reason that emollients are recommended for burn patients after healing. Emollients increase the water content of the stratum corneum (outer layer of skin), filling the spaces between partially desquamated skin flakes, and so the skin appears smoother (Klotz et al, 2017). Itching is a common side effect of hypertrophic burn scars and can be very problematic for patients, but hydration of the skin with emollients has been shown to reduce this itch (Chung et al, 2020). Often patients are advised to keep their emollients in the fridge, because cooling the scar can also help to reduce itch (Chung et al, 2020). Ideally, emollients need to be free from fragrances, colours, preservatives and sodium lauryl sulphate (SLS), and minimum amounts should be used, because too much can block new pores and cause breakouts or build up of a film of emollient (Klotz et al, 2017).
Massage
Massage is often commenced once the healed tissue has gained sufficient strength to tolerate surface friction, and it is facilitated by lubricants (such as moisturisers/emollients). A survey of burn therapy practices identified that 81% of responding units routinely advocated scar massage as a preventative therapy (Holavanahalli et al, 2011). Ault et al (2018) demonstrated that massage can improve pain, itch, scar height, pliability and vascularity. All patients should be taught how to massage once the wound has healed, gently at first, then increasing in pressure until they are making the scar blanch. There is no agreement on the exact method of massage, but generally it is advocated to be done two to three times a day and more if early signs of hypertrophy appear (Figure 4).
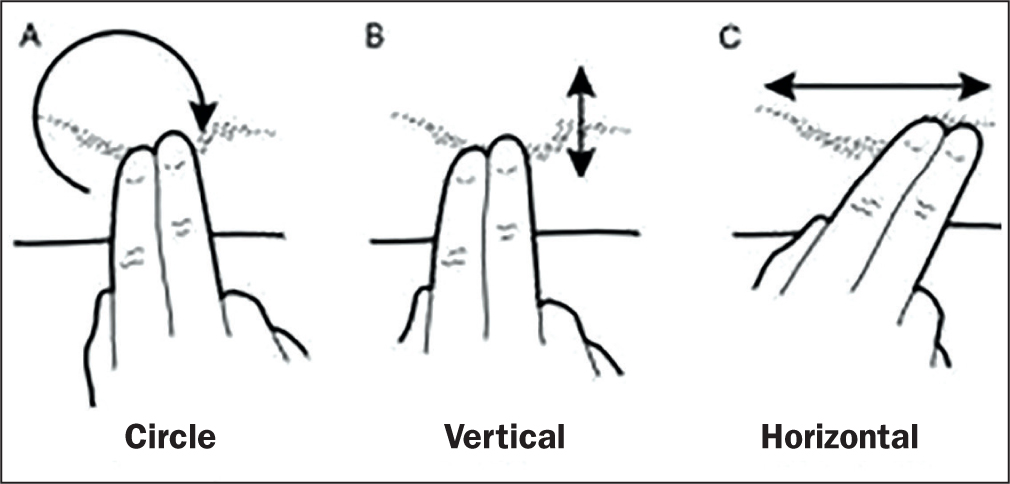
Ultraviolet-light protection
Due et al (2007) demonstrated that ultraviolet radiation increases scar pigmentation and worsens their clinical appearance. Middelkoop et al (2011) added that avoidance of exposure to sunlight is key and that sun cream with a sun protection factor (SPF) of at least 50 should be used until the scar has matured (usually in 2 years). In clinical practice, it is worth using children's sun cream, as this rubs in more easily on delicate newly healed skin. This should be reapplied regularly, as the blocking effect only lasts for 2 hours (MacGill, 2018).
Scar treatment with silicone therapy
Since the early 1980s, silicone gel has been used to manage hypertrophic scars (Teot et al, 2020a). Over 60 different silicone gels have been marketed since 1990, so there is a lot of choice available to the practitioner (Friedstat and Hulman, 2014). The exact mechanism of action is still not fully understood, but numerous mechanisms have been suggested to explain the efficacy of silicone. These include hydration, pressure, temperature, oxygen transmission and silicone absorption (Momeni at al, 2009). Of these, hydration seems to have the most evidence to support it. Covering a scar with silicone leads to the accumulation of water in the stratum corneum, resulting in hydration through the formation of a water reservoir (Hoeksema et al, 2013). Of many scar treatments, silicone is thought to have the most clinical evidence of its efficacy; however, a recent Cochrane review into silicone gel sheeting found that there was not enough high-quality evidence of their efficacy (Jiang et al, 2021). Clinically, however, most scar clinicians see a positive outcome from its use, so it is still considered a first-line treatment.
Parry et al (2013) suggested that silicone is associated with improved scar elasticity, decreased hypertrophy, scar roughness and itch. They added that silicone may influence the scar later in scar development, and, certainly clinically, there have been positive results from silicone therapy on scars up to 10 years of age. Silicone therapy must be increased gradually, or rash/irritation can occur. Therefore, it is usually applied for 2 hours the first day, increasing daily by 2 hours until reaching 8-10 hours, when it can be then worn for up to 24 hours a day. It must be removed daily to allow for washing of the scar, and many silicone gel sheets also need washing. If this is not done, then there can be issues with itching, rashes, maceration and odour (Teot et al, 2020a). Silicone therapy is considered a first-line treatment for hypertrophic scars.
Conclusion
Management of the skin following healing of the burn wound is key to a good cosmetic and functional outcome. Preventive scar management will help to prevent the development of hypertrophic scars, which could lead to significant functional and psychological difficulties, and it is achievable by almost all practitioners.
The simple act of advising the use of emollients, massage and sun protection is able to make a significant difference to any patient with a burn wound. Silicone therapy may not be for all non-specialised practitioners, but, for those who have the knowledge and confidence to use it, it would make a good first-line treatment and is available on prescription. If this is not effective, then it would be appropriate to refer the patient to a burn service for more intensive scar therapies.
KEY POINTS
- Hypertrophic scarring can develop after any wound, but is more common in burn injuries
- Hypertrophic scarring usually occurs within 4–6 weeks post-wound healing
- Simple massage can help to prevent hypertrophic scarring
- Nurses are ideally placed to advise patients on scar prevention
CPD reflective questions
- Why does hypertrophic scarring develop?
- What impact does hypertrophic scarring have on the patient?
- What simple advice can nurses give to patients to prevent hypertrophic scarring?


