Arange of antiretroviral therapies (ART) is available to treat HIV infection, preventing onward transmission and new diagnoses, and reducing mortality. However, to be effective, these treatments require a person living with HIV to take a combination of oral tablets every day for the rest of their lives to ensure that HIV viral load levels are suppressed to a low enough level for these to be considered undetectable.
In the UK, 97% of people living with HIV are on therapy and have an undetectable viral load (Delpech, 2022). This allows them to live well with a similar life expectancy to the rest of the population. However, treatment success relies on good adherence. Since the advent of triple ART therapy in 1996, research has successfully led to the introduction of fixed-dose combinations, single-tablet regimens and once-daily scheduling, all of which has made adhering to therapy less of a burden. The medications are also more tolerable with fewer long term sideeffects. Nonetheless, for many patients swallowing medication at a set time every day is challenging, and suboptimal adherence can potentially lead to virological failure with subsequent resistance, requiring a switch in treatment, which often requires more tablets and twice-daily scheduling (British HIV Association (BHIVA), 2023).
The introduction in 2021 of long-acting injectable ART has provided an alternative option for those people living with HIV who can no longer tolerate oral medication. Pooled analysis of clinical trials led to Scottish Medicines Consortium (SMC) (2021) approval of long-acting ART, in the form of two injectable antiretroviral drugs, for use in Scotland, followed by a recommendation by the National Institute for Health and Care Excellence (NICE) (2022). Both the BHIVA and European AIDS Clinical Society (EACS) have developed guidelines for consideration of these treatments (BHIVA, 2023; EACS, 2023).
The formulations allow for continuous drug exposure that lasts over the duration of the dosing interval. Rilpivirine (RPV), a non-nucleoside reverse transcriptase (NNRTI), and cabotegravir (CAB), an integrase inhibitor, are injected into a different muscle and are not given in combination with any other oral antiviral medications.
Evidence
The key clinical evidence for long-acting CAB/RPV came from the ATLAS (Antiretroviral Therapy as Long Acting Suppression), FLAIR (First Long-Acting Injectable Regimen) and ATLAS-2M studies (Swindells et al, 2020; 2022; Orkin et al, 2021). These were phase 3 randomised, controlled, open-label, non-inferiority trials in people living with HIV-1.
ATLAS (Swindells et al, 2020) and FLAIR (Orkin et al, 2021) demonstrated that long-acting CAB with long-acting RPV is non-inferior to oral ARTs, with the primary outcome of viral suppression achieved in both trials. ATLAS-2M (Jaeger et al, 2021) showed that long-acting CAB with long-acting RPV was non-inferior when administered every 2 months compared with being given every 1 month.
In the ATLAS-2M study (Swindells et al, 2022), 98% of patients, when surveyed at week 48, said they preferred having an injection every 8 weeks compared with taking a daily oral tablet. The 52-week results of the same study also illustrated patient preference for injections 2-monthly versus every month (Overton et al, 2023).
The clinical trials (Jaeger et al, 2021; Orkin et al, 2021, Swindells et al, 2022; Overton et al, 2023) showed that the injectable treatment was safe and effective in people living with HIV, but these studies included only a highly select group of patients, in that these were individuals who had been completely adherent to their oral therapy regimen and had maintained viral suppression.
The trials showed that, despite complete adherence to injectable therapy, complete virological failure (CVF) was seen in a small group of participants (Orkin et al, 2023). The risk of failure in ATLAS 2M was 1 in 70 after 1 year, 1 in 60 after 2 years and 1 in 40 after 3 years, and most failures resulted in resistance to two classes of ART.
The rate of CVF was modestly increased by the presence of at least two baseline factors:
- Archived RPV resistance-associated mutations, eg in cases of incomplete treatment and viral resistance history
- HIV-1 subtype A6/A1 (uncommon outside the Russian Federation)
- BMI >30kg/m2.
No single baseline factor alone was predictive of virological failure. Real-world data show that virological failure has less frequent treatment-emergent resistance (Kityo et al, 2024); however, it is vital that patients are fully informed of the risks of virological failure before making a decision to start injectable therapy. People living with HIV should always be actively involved in decisions about their own health (BHIVA, 2018).
Due to the risk of developing dual-class resistance, BHIVA issued strict guidelines to ensure that patients being considered for injectables had excellent adherence, had maintained an undetectable viral load for at least 6 months and had no previous resistance or treatment interruptions. It is also important to note that injectables are licensed only for people over the age of 18 years and cannot be used in pregnancy or for patients co-infected with hepatitis B.
Due to the risk of virological failure on injectables, BHIVA (2023) guidelines require there to be a significant need for this treatment option, with the following patient factors taken into account when assessing an individual's suitability for injectables:
- Major psychological impact of daily pills
- Physical barriers to pill taking
- Viral load undetectable, despite concerning adherence
- Expressed risk of stopping ART if expected to continue oral ART.
Benefits of an injectables pathway
Concerns about daily medication
Holistic care highlights the need to improve quality of life as people with HIV are now living longer. Effective medication should allow someone to remain well and have a healthy and happy fulfilling life, but there are many barriers to enabling this (Figure 1).
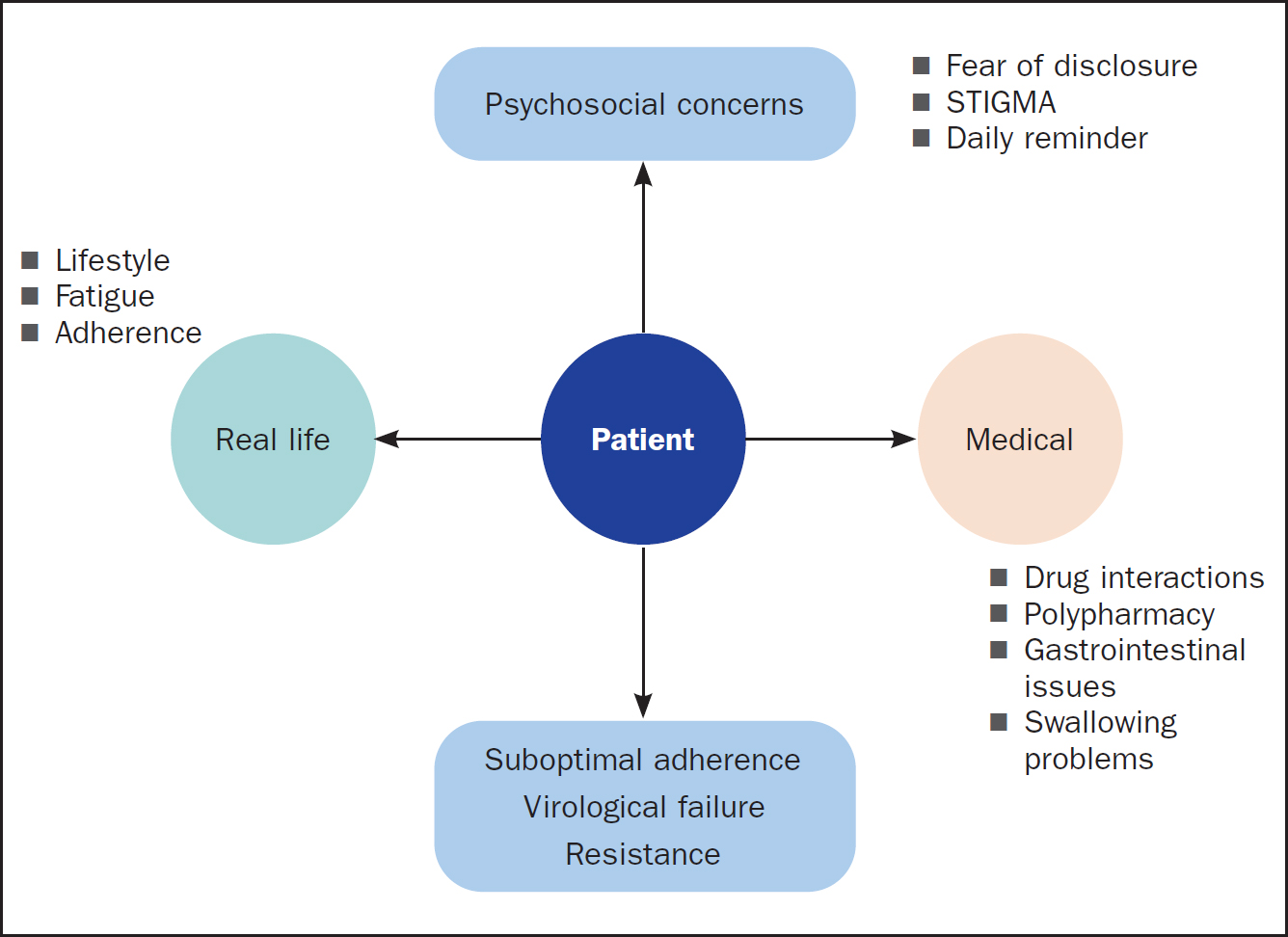
Stigma is often a factor that prevents people living with HIV disclosing their HIV status to friends or loved ones, and so they will feel the need to hide their tablets. Some individuals become extremely anxious about keeping up with adherence or the consequences of missing a dose of their antiviral medication, others dread the daily reminder of living with HIV every time they swallow their medication, while others may simply have pill fatigue from years of taking lifelong daily treatment. In other cases, an individual's lifestyle can make it difficult to adhere to the medication schedule; for example, they may have a busy job that involves travelling outside the UK or that may have frequently changing shift pattern. Mothers tend to prioritise their children's health and forget to self-manage their own.
As patients live longer, they become at higher risk of developing chronic comorbidities resulting in polypharmacy, which increases the possibility of drug-drug interactions with antiviral medication. A range of medical problems can also make swallowing daily medication challenging. As patients age, they may also develop cognitive problems and may forget to take their tablets.
Some patients who have taken therapy for many years simply reach the point of feeling that they cannot continue to take the medication every single day. In each of these cases, there is the possibility of suboptimal adherence, which can lead to treatment failures and subsequent poor health.
How an injectable pathway works
A multidisciplinary discussion is held to ensure an eligible patient fits all criteria. They are then signposted to the injectable service. In the majority of units around the UK, this is a nurse-led service. At the first visit, the patient meets the nurse and the pharmacist, who fully inform them of the procedure and the pathway of care, and the pharmacist discusses any potential side effects. Guidelines no longer include the 4-week oral lead-in that involves taking cabotegravir 30mg and rilpivirine 25mg in oral form. However, many care providers still include this step, because it ensures that a patient is able to tolerate the medication. This pathway is followed by the author's Trust, NHS Lothian. It is important that patients are counselled to take rilpivirine with a main meal because at least 400 calories are required in order for it to be absorbed correctly.
If using the oral lead-in (Figure 2a), the first injection is administered on the final day of oral treatment. Subsequently, two deep right and left ventrogluteal intramuscular (IM) injections are administered: at 1 month and at 2 months after the initial injection. Thereafter, two deep IM gluteal injections are administered every 2 months in clinic. Bloods are taken at each visit with prompt recall for repeat testing and resistance testing if viral rebound occurs. An annual review is carried out every 12 months for a holistic physical, psychological and social wellbeing assessment.
The alternative pathway without oral lead-in is illustrated in Figure 2b, and follows the same pattern of appointments as the oral lead-in pathway, except that the patient has not taken oral medication for an initial 4 weeks.

Drug interactions
If using the oral lead-in, antacids including calcium or magnesium and multivitamins must be taken 2 hours before and 4 hours after taking oral cabotegravir. Antacids such as famotidine, lanzoprazole, omeprazole and ranitidine should be avoided when taking oral rilpivirine.
Long-acting CAB/RPV injections bypass the gut, therefore this route of administration avoids many drug interactions related to oral absorption. There are no food requirements, no pH constraints and no binding to polyvalent cations; however, there are still certain medications that cannot be taken with long-acting CAB/RPV, which include:
- Anticonvulsants used to treat epilepsy and prevent seizures (carbamazepine, oxcarbazepine, phenobarbital, and phenytoin)
- Antibiotics to treat some bacterial infections, for example tuberculosis (rifapentine, rifampicin, and rifabutin)
- Dexamethasone, unless a single-dose treatment
- St John's wort (Hypericum perforatum).
Caution should be used when long-acting injectables are taken with some antibiotics, namely clarithromycin, erythromycin and flucloxacillin. As more real-world data are obtained, more potential interactions may emerge, therefore drug interaction checks should be part of routine follow-up. A good source of information on this aspect is the University of Liverpool HIV Drug Interactions website (https://www.hiv-druginteractions.org/checker).
Injectables are safe with gender-affirming hormones and contraception (see https://www.hiv-druginteractions.org/checker).
Considerations when setting up an injectable clinic
Patient assessment
A thorough assessment of a patient is necessary to ensure that the eligibility criteria have been checked and the MDT is in agreement to commence long-acting injectables. The patient should undergo the necessary counselling to ensure that there is an understanding of the potential risk of virological failure. The patient needs to agree to attend at the allocated appointment every 8 weeks and be aware of the importance of avoiding missing appointments. The increase in the frequency of visits has an impact on service capacity and some clinics choose a specific day of the week to deliver this service. Clinics need to have the capacity to provide space for patients to be observed post procedure and carry out phlebotomy at each visit. It is also important that there is refrigeration capacity for the number of patients and that cold chain requirements can be adhered to. Staff need to be trained and supported to feel competent to carry out IM ventrogluteal injections.
There must be robust pathways in place to ensure that patients who virally rebound or miss appointments are recalled in a timely manner. There is a ±7 day window period for early/late administration and oral bridging can be used, but should be considered only as an exception rather than used routinely.
Injection site reactions (ISRs)
Ring et al (2023) carried out a UK-based service evaluation in 2023 to look at the discontinuation rate. The rate was low and mainly due to low level viraemia or virological failure. Some patients struggled with the 2-monthly clinic visits and others found the injection site reactions too painful, but there were less than 1% reported serious injection-site reactions (ISRs).
The most common injection-site reactions are:
- Pain
- Swelling
- Headache
- Feeling hot
- Pyrexia.
In the ATLAS, FLAIR and ATLAS 2M studies (Swindells et al, 2020; Orkin et al, 2021; Overton et al, 2023) less than 2% of discontinuations was due to injection-related reasons, with 98% of ISRs recorded as mild to moderate, and that these decreased over time. People reported experiencing various degrees of pain at injection sites, with discontinuations occurring if pain became more than mild to moderate, and persisted for longer than 3–4 days. Other reactions, such as abscesses or developing liver issues, also resulted in discontinuation.
Patient feedback
For some people living with HIV, the disadvantages may outweigh the benefits. Attending every 8 weeks rather than twice a year could be inconvenient, especially if this involves travelling a long distance. Others may be anxious that extra appointments could be questioned by employers or friends and family. In other cases, patients may not like the concept of deep painful IM injections, or may question whether two injections every 8 weeks can be as efficacious as daily oral treatment.
However, feedback from patients receiving injectable therapy has been very positive (Murray et al, 2020).
Uptake and future use
There was initial excitement from patients, clinicians and the voluntary sector when long-acting injectables became a treatment option for people living with HIV. However, the number of potential candidates has reduced due to both clinicians and patients being wary of the virological failure rates despite adherence to injectable dosing schedules. This is pertinent for those patients who rely on the U=U statement, namely that people who remain on therapy and sustain an undetectable viral load cannot transmit HIV to their sexual partners (Rodger et al, 2019).
The strict eligibility criteria mean that only those patients who are able adhere to oral medication and achieve undetectability for the previous 6 months are considered for the injectables treatment option. There have been exceptions for patients who have run out of treatment options, and applications can also be made for off-licence prescribing on compassionate grounds.
It is important to acknowledge that, because injectables have been investigated only in the context of viral suppression in a highly select population, currently no data are available on the efficacy of this treatment option in more complex patient populations, including those with detectable viral loads due to poorer adherence. Patients with substance use, mental health issues, prisoners and those affected by homelessness could all benefit from this form of treatment. A study in Australia by John et al (2023) illustrated that people living with HIV and identified as having complex needs, social vulnerability and poorer adherence patterns achieved the same levels of adherence and virological suppression as the wider population in the study, due to enhanced intensive support from social workers, drug workers and specialist nurses.
Patients who struggle with oral adherence due to mental health issues or substance use may well manage to come to clinic every 8 weeks and could do better on this therapy because they do not have to remember to take a daily tablet. Young adults may also struggle to remember to take daily oral medication and attend clinic because of their busy lives, so could similarly benefit from injectables. However, clinics will need to be inventive and consider holding evening clinics, if HIV services require them to attend for more frequent appointments for administration.
More collaborative working between community support agencies and HIV services is needed to help patients attend more frequent appointments. In the current financial climate, HIV services need to look at novel, more cost-effective ways of delivering care to people living with HIV. Consideration also needs to be given to support the concept of other clinical departments outwith the HIV setting providing a service to deliver injectables. Although long-acting CAB and RPV treatment is an expensive option, the long-term consequences of poor adherence, which inevitably require more expensive regimens and potentially the need for hospital admissions if the patient becomes clinically unwell, must be taken into consideration.
Conclusions
The introduction of long-acting injectable HIV therapy has been life-changing for those people living with HIV who needed an alternative option to taking oral medication. For many, swallowing ART is a constant reminder of the stigma they face living with HIV. For others, it could be that they have swallowing problems or gastrointestinal issues, and long-acting injections provide an alternative delivery route.
Clinical trials have shown that injectable treatment is safe and effective in people living with HIV. However, these trials have included a highly select group of patients and, despite complete adherence, certain patients had complete virological failure and developed two-class resistance. The CARES study in Africa (Kityo et al, 2024), however, has shown that long-acting CAB and RPV showed non-inferior efficacy compared to standard oral ART and, despite many patients having pre-existing rilpivirine resistance and subtype A1 virus, there were very few complete virological failures or acquired resistance. Further research is needed to investigate the reasons for this in order for it to be considered a viable option for Sub-Saharan countries.
Meanwhile, in the UK, BHIVA treatment guidelines (BHIVA, 2023) advise that there must be a significant clinical need before choosing this treatment option and most HIV centres hold their own MDT meetings prior to making such clinical decisions.
Nurses are leading the way in many chronic conditions and HIV is no exception. HIV nurses have the skills to develop protocols and pathways to deliver this new treatment model. It is the role of the HIV nurse to deliver the highest quality of care and provide the best treatment options enabling people living with HIV to lead healthy fulfilling lives.
KEY POINTS
- Good adherence to antiretroviral therapy can ensure that a person living with HIV can maintain an undetectable viral load and avoid onward transmission of the virus
- Long-acting injectable antiretroviral therapies (long-acting ART) is an option for people living with HIV who are not able to take oral medication
- HIV nurse specialists are ideally placed to develop a nurse-led service to ensure the safe delivery of long-acting injectables
CPD reflective questions
- What are the benefits of long-acting injectables?
- What are the challenges of implementing long-acting injections in your clinics?
- Can you think of other patients who may benefit from this type of treatment who currently do not fit the eligibility criteria?


