Approximately 700 000 people in Europe and more than 1 million in the USA live with an ostomy (Claessens et al, 2015). Although stoma formation can be lifesaving for patients (Burch, 2005), it may also compromise physical and psychosocial wellbeing and lead to reductions in quality of life (QoL) (Claessens et al, 2015; Richbourg et al, 2007).
Leakage of stomal effluent is one of the key factors leading to reduced QoL (Davis et al, 2011; Claessens et al, 2015), especially for people experiencing leakage onto clothes (Jeppesen et al, 2022). Besides being socially embarrassing, it is distressing and inconvenient for the individual to experience leakage of stomal effluent outside the baseplate (eg, onto clothes or bed sheets) and leakage of effluent onto the skin is associated with the development of peristomal skin complications (Voegeli et al, 2020). More than 90% of people with an ostomy worry about leakage to varying degrees (Claessens et al, 2015; Jeppesen et al, 2022). The reportedly high rates of those worrying about leakage suggest that current ostomy care products do not address the issue effectively.
The aim of this prospective, single-arm, pilot study was to evaluate performance of a novel digital leakage notification system (Heylo). Heylo is a CE-marked (approved for European markets) adherent medical sensor device that, when attached to an ostomy baseplate, constantly measures the status of the user's baseplate and, via Bluetooth communication, sends status changes to a bespoke smartphone application. The purpose of the product is to help those with intestinal stomas to gain better control of their ostomy care by letting users know when effluent is seeping underneath the baseplate. The notification system potentially alerts users to take action to avoid leakage progressing outside the baseplate.
The innovation consists of four elements, as shown in (Figure 1). The first is a sensor layer, with two circular leakage sensor rings, that monitors the interface between skin and adhesive for moisture as a sign of leakage. The inner sensor ring consists of three zones and the outer ring of a single zone, allowing the system to monitor both leakage direction and progression (Figure 2). A transmitter is attached to a connector on the sensor layer, enabling readout of the individual sensor and is used to calculate the status of the sensor layer. The smartphone app displays the status of the baseplate to the user and ‘pushes’ notifications in case of relevant changes. The app can inform the user: ‘Looking good’, ‘There is a problem, it may be leakage’, ‘The problem is spreading’ and so on. A transmitter charger is delivered with the system.

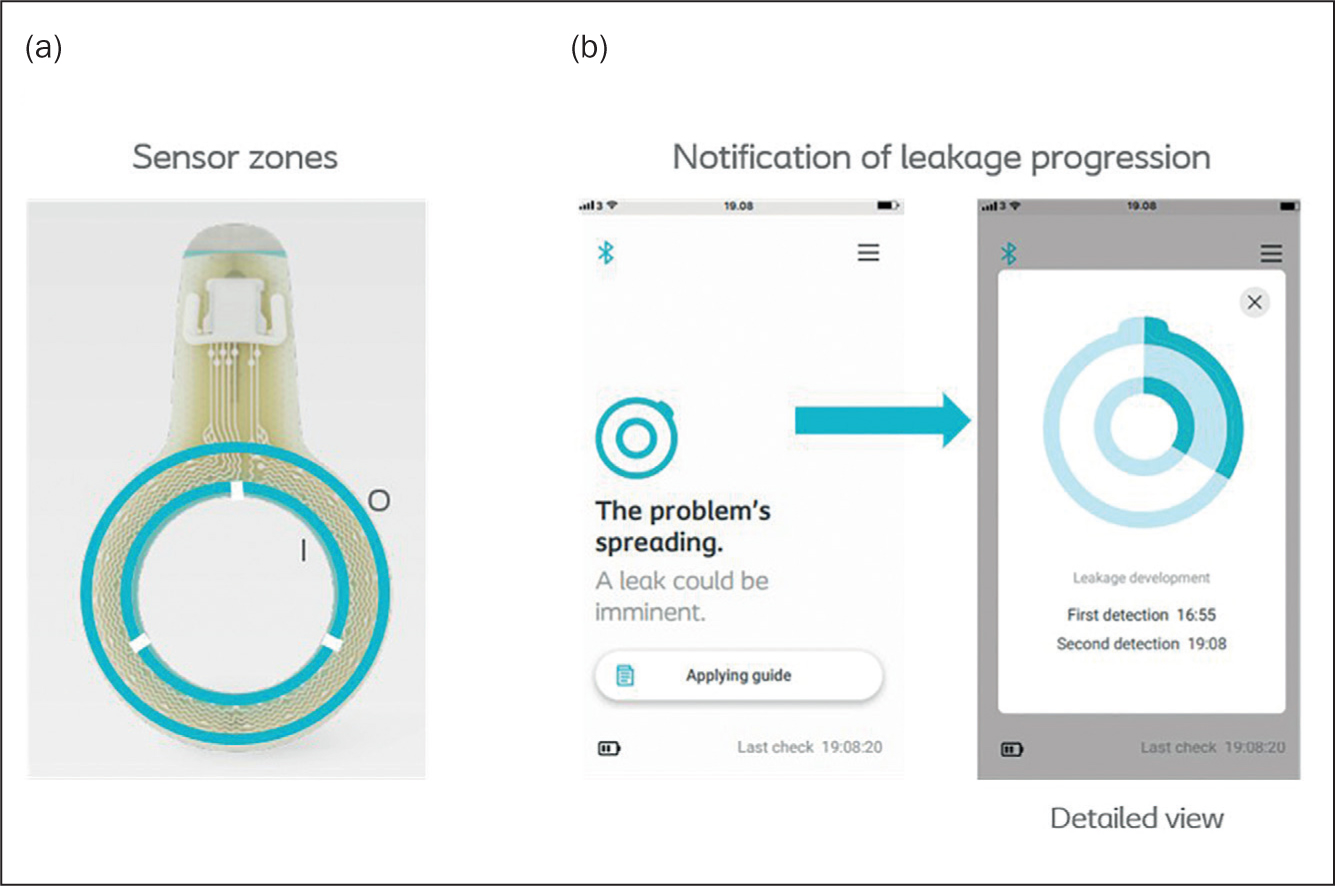
This article reports on system performance, users' experience of the system, the system's ability to prevent leakage progressing onto clothes, and the impact on worry about leakage and QoL.
Method
Study design
The study was a single arm, open-label, exploratory investigation comprising 25 subjects assigned to use of the test product (Heylo) for 21±3 days. Three virtual visits were scheduled: an inclusion visit (V0), a visit for introduction and onboarding of patients to the test product at day 1 (V1), and a termination visit (V2). Baseline demographics were collected at V1 and questionnaires were filled in at both V1 and V2. Subjects were trained in use of the test product and how to fill in the questionnaires. Time between inclusion visit (V0) and onboarding visit (V1) may have been up to 1 week, as products were shipped by post to subjects.
Test product
Subjects were provided with a smartphone with the bespoke Heylo app preinstalled, as well as an extra transmitter, and enough sensor layers for users to change effluent collection systems to a similar routine as they would do normally. The smartphones included in this investigation were iPhones (X, XS) using the iOS operating system (n=10), and Samsung phones (S7 and S9) using the Android operating system (n=15).
Selection of study participants
Subjects were recruited by sending invitation letters to people with an ostomy already registered in Coloplast A/S databases, and who had given consent to receive information regarding future participation in clinical investigations. Inclusion criteria identified those with an ileostomy or colostomy being over 18 years of age, who had had their stoma for longer than 3 months and had mushy to liquid output (Bristol scale type 6 or 7) (Lewis and Heaton, 1997). Subjects could use a flat, convex, or concave one-piece or two-piece pouching system of a Coloplast A/S brand (Assura, SenSura or SenSura Mio). Subjects had to have a minimum of three episodes of leakage underneath the baseplate within the previous 14 days and had to worry about leakage to ‘some’, ‘high’ or ‘very high’ degree (based on a five-point Likert scale).
Exclusion criteria involved those with severe peristomal skin complications (minor discolouration of the peristomal skin was allowed), people receiving topical or systemic steroid treatments, or people undergoing radiotherapy or chemotherapy treatment. The investigators hypothesised that steroid treatment or radiotherapy/chemotherapy may compromise the skin condition and therefore bias study outcomes. People with sensitivity to acrylate, people using ostomy paste or powder, and females being pregnant or breastfeeding were excluded from participating in the study.
Patient demographics and endpoints
Demographics and patient variables (gender, stoma type, years with stoma, reason for stoma surgery etc) were recorded at baseline (V1).
Primary endpoint
Following each baseplate removal users took pictures showing the sensor area. Each image was assessed for leakage in the sensor areas by two people from the clinical study team. If leakage was observed, the sensor layer was considered as having a positive incident (Figure 3). If no leakage was observed, the sensor layer was considered a negative event. In case of disagreement, the result was aligned in a review meeting with several participants. In some cases, the image quality was at a level where assessment could not be made, and the baseplate was removed from evaluation. The complete evaluation of images was used as a reference dataset to calculate accuracy of how well the system detected leakage.
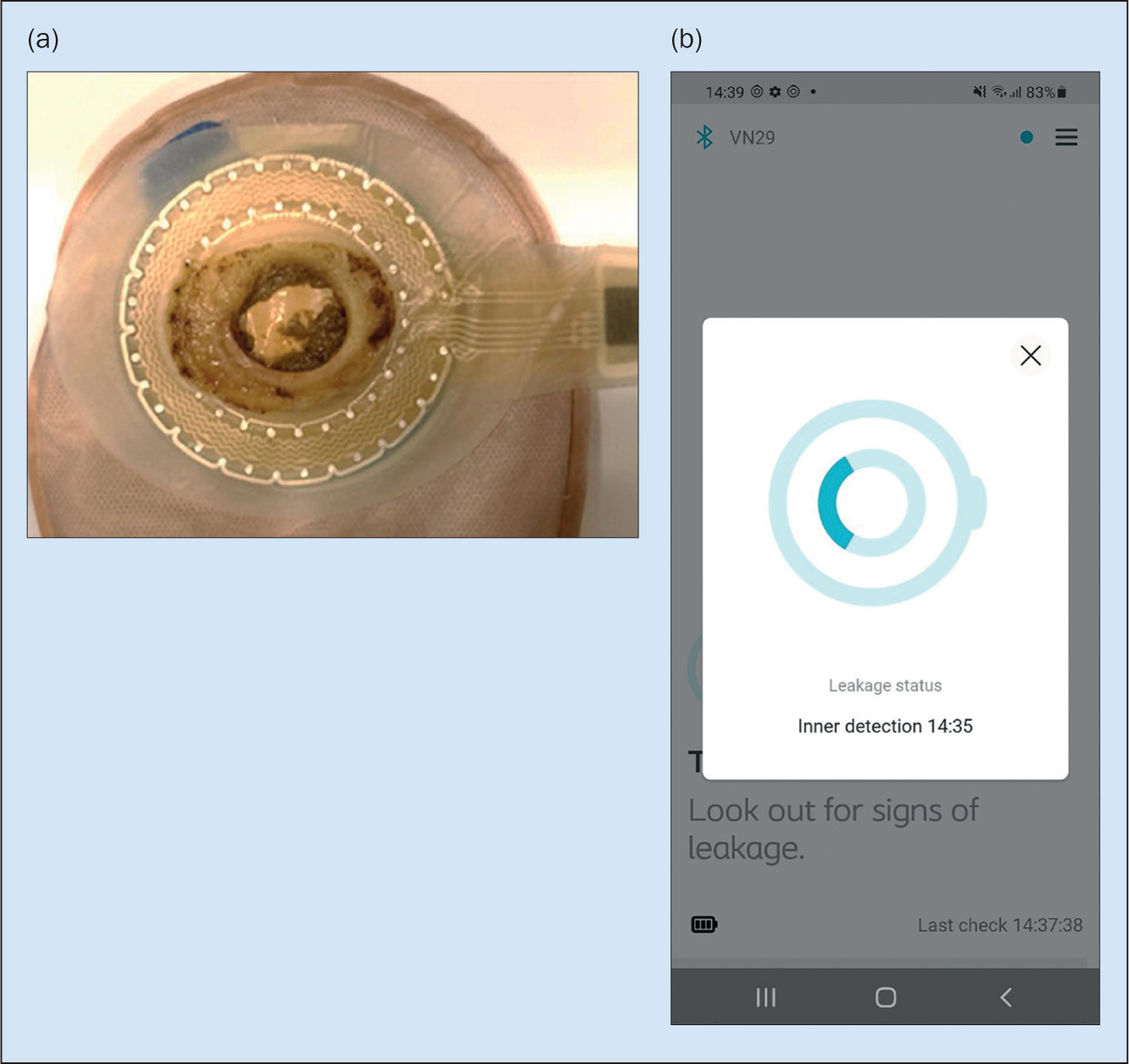
To evaluate whether the system was predictive, the notification status was extracted at the nearest datapoint prior to the timestamp of the picture. If the system detected leakage, it was considered a positive event, in contrast it was considered a negative event if the system did not detect leakage. The reference dataset was then compared against the system dataset with the following potential outcomes:
- True negative: No leakage observed on picture and no leakage detected by system
- True positive: Leakage observed in picture and leakage detected by system
- False negative: Leakage observed in picture, but no leakage detected by system
- False positive: No leakage observed in picture, but leakage detected by system.
Performance accuracy was calculated accordingly as: ((True positive+True negative)/(True positive+True negative+False positive+False negative))×100
Secondary endpoints and other assessments
At baseline (V1) and at termination of the study (V2), subjects were asked to:
- Report the number of times they had experienced leakage onto clothes during the previous 21 days
- Describe their level of worry about stoma-related leakage within pre-defined options on a five-point Likert scale (‘very low degree/not at all’, ‘low’, ‘some’, ‘high’ and ‘very high’ degree)
- Evaluate their burden of leakage using the validated Ostomy Leak Impact (OLI) tool (Nafees et al, 2018). The OLI tool summarises the burden of leakage in three domains: ‘emotional impact’, ‘usual and social activities’ and ‘coping and control’
- Complete 23 questions from the Ostomy-Q tool, a tool designed to measure the QoL related to aspects of using an ostomy appliance (Nafees et al, 2017). The questions are grouped in four domains, concerning discreetness of the ostomy appliance, comfort related to wearing the ostomy appliance, confidence with the ostomy appliance, and how the appliance impacts social life and relationships (Nafees et al, 2017).
At termination of the study (V2), subjects were also asked to evaluate:
- To which degree did you find the notifications from the app reliable and trustworthy?
- To which degree did the test product make it easier to know when to change baseplate compared with your usual product?
- Did the test product give you a feeling of leakage control compared with your usual product?
- Did the test product give you a feeling of control of leakage progression compared with your usual product?
- Compared with your usual product, did the test product affect your sleep?
At each baseplate change subjects evaluated:
- What was the reason for baseplate change?
Questions could be answered from lists with pre-defined options, which are shown in the figures and tables. Adverse events were recorded continuously throughout the study.
Data management and statistics
The sample size for this pilot study was estimated based on previous experience with system performance (Coloplast A/S internal data). Statistical analyses were performed using SASv9.4 (SAS Institute Inc, Cary, NC) after data entry and data management using a validated data management system (Rave EDC, v2018.2.2, Medidata Solutions).
The comparison of number of leakages onto clothes from baseline (V1) and at study termination (V2) were analysed using a Poisson loglinear model, whereas worry about leakage was analysed by a proportional odds ratio model. The domain scores and total score of Ostomy-Q, as well as the domain scores of the OLI tool, were analysed by paired T tests. Questions answered on two-point Likert scales and questions answered on five-point Likert scales (the answers were subsequently assigned to two groups) were analysed by use of an exact test in the binomial distribution. It was tested if the group evaluating the test product positively was significantly different from 50% (H0: π=0.5; α=0.05). If the lower confidence limit was above 50%, this meant that the test product was assessed positively by most users.
Ethical consideration
The study was carried out in accordance with the Declaration of Helsinki and was approved by the Ethical Committee of the Capital Region in Denmark as well as by regulatory authorities (Danish Medicines Agency, https://laegemiddelstyrelsen.dk) before study initiation. The study was registered on ClinicalTrials.gov (NCT04894084). All subjects were fully informed about the investigation, both verbally and in writing, and all gave written informed consent to participate in the study. Participation was voluntary and subjects could withdraw at any time. The subjects received a small gift voucher for participation in the study. It was conducted from April to July 2021 in Denmark.
Results
Demographics of study participants
Twenty-five subjects were included in the study: 15 (60%) had an ileostomy and 10 (40%) had a colostomy. The study population consisted of 10 females (40%) and 15 males (60%). The average age of the subjects was 55.9 years (range 36-80; SD=11.9) and subjects on average had had their stoma for 11.8 years (range 1-34; SD=8.4) (Table 1).
Table 1. Baseline characteristics of study population
| Parameter | Total n=25 |
|---|---|
| Age (years): Mean ± SD (range) | 55.9 ± 11.9 (36;80) |
| Sex: n (%) | |
| Females | 10 (40) |
| Males | 15 (60) |
| Years since stoma creation: Mean ± SD (range) | 11.8 ± 8.4 (1;34) |
| Type of stoma: n (%) | |
| Ileostomy | 15 (60) |
| Colostomy | 10 (40) |
| Reason for stoma creation: n (%) | |
| Ulcerative colitis | 7 (28) |
| Cancer | 6 (24) |
| Crohn's disease | 5 (20) |
| Other | 7 (28) |
| Consistency of stool: n (%) | |
| Bristol Scale 6 | 22 (88) |
| Bristol Scale 7 | 3 (12) |
Seven (28%) subjects had stomas created due to ulcerative colitis, 6 (24%) due to cancer, 5 (20%) due to Crohn's disease and 7 (28%) due to other causes that were not specified (Table 1). All but two subjects used SenSura Mio products (Coloplast A/S) as their regular ostomy care product, and the remaining two used SenSura products (Coloplast A/S). Eleven (44%) used one-piece and 14 (56%) used two-piece products. Most subjects used either a flat (n=12, 48%) or a convex (n=10, 40%) product, and the remaining subjects used a concave product (n=3, 12%).
One subject withdrew prematurely from the study (at 10 days) after having technical difficulties with the system, but nonetheless completed the termination questionnaire (V2 moved forward and was included in the analysis of V2 data). All subjects reported at the screening interview (V0) that they worried to ‘some’ or higher degree about leakage. However, when filling in the questionnaire at baseline (V1), two subjects reported only a ‘low’ degree of worry about leakage. Since the subjects reported ‘some’ degree of worry about leakage at the screening interview (V0), and thus were already enrolled, it was decided that data from these two subjects should still be included in the final analysis and, as such, was not assessed to be a protocol deviation. After study completion, questionnaire data from V2 could not be retrieved from two of the subjects (due to a technical issue with the clinical trial app on the phones), hence follow-up data included in the final comparative analysis was based on responses from 23 subjects. Data collected at V1 from all subjects (n=25) have been included in the analysis on a cross-sectional basis. Results from the intention-to-treat (ITT) population are presented in the following sections.
Performance accuracy
A total of 360 baseplate changes were recorded during the test period and images for 317 of those were of sufficient quality to assess performance accuracy. Performance accuracy was 78% (95% CI (74%; 83%)), suggesting high concordance between messages conveyed to the user and baseplate pictures. In 22% of the changes, the status message did not match with the leakage picture. Issues leading to mismatches include software bugs, loss of Bluetooth connection between transmitter and phone, and early depletion of sensors due to mounting on wet skin that is not compliant with the intended use.
Subject evaluation of performance
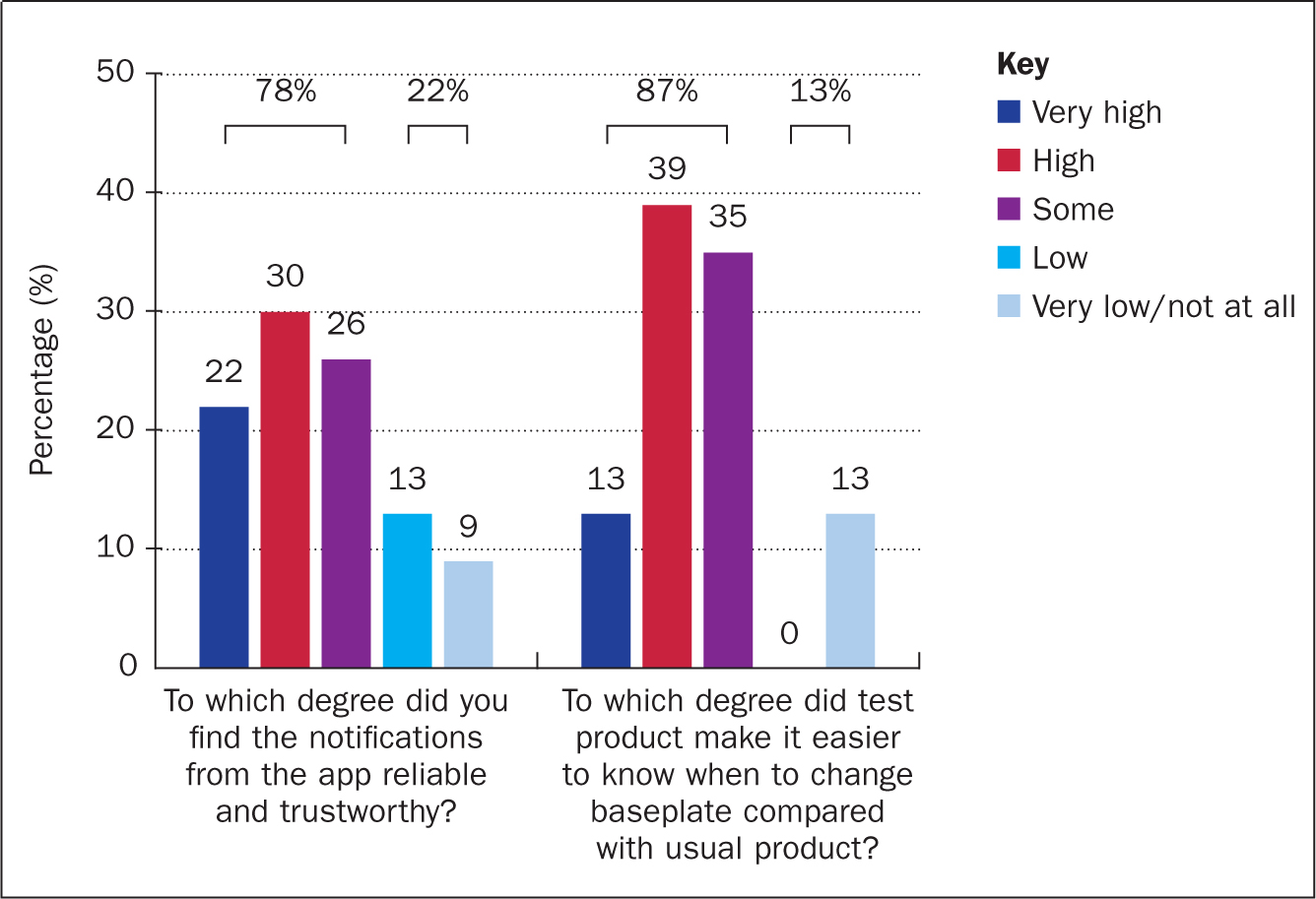
Eighteen subjects (78%, 95% CI (56%;93%), P=0.011) stated that notifications were reliable and trustworthy to ‘some’ (26%), ‘high’ (30%) or ‘very high’ (22%) degree (Figure 4). Twenty subjects (87%, 95 CI (66%;97%), P<0.001) stated that the test product to ‘some’ (35%), ‘high’ (39%) or ‘very high’ (13%) degree made it easier to know when to change baseplate compared with only using their own usual product (Figure 4).
Leakage onto clothes
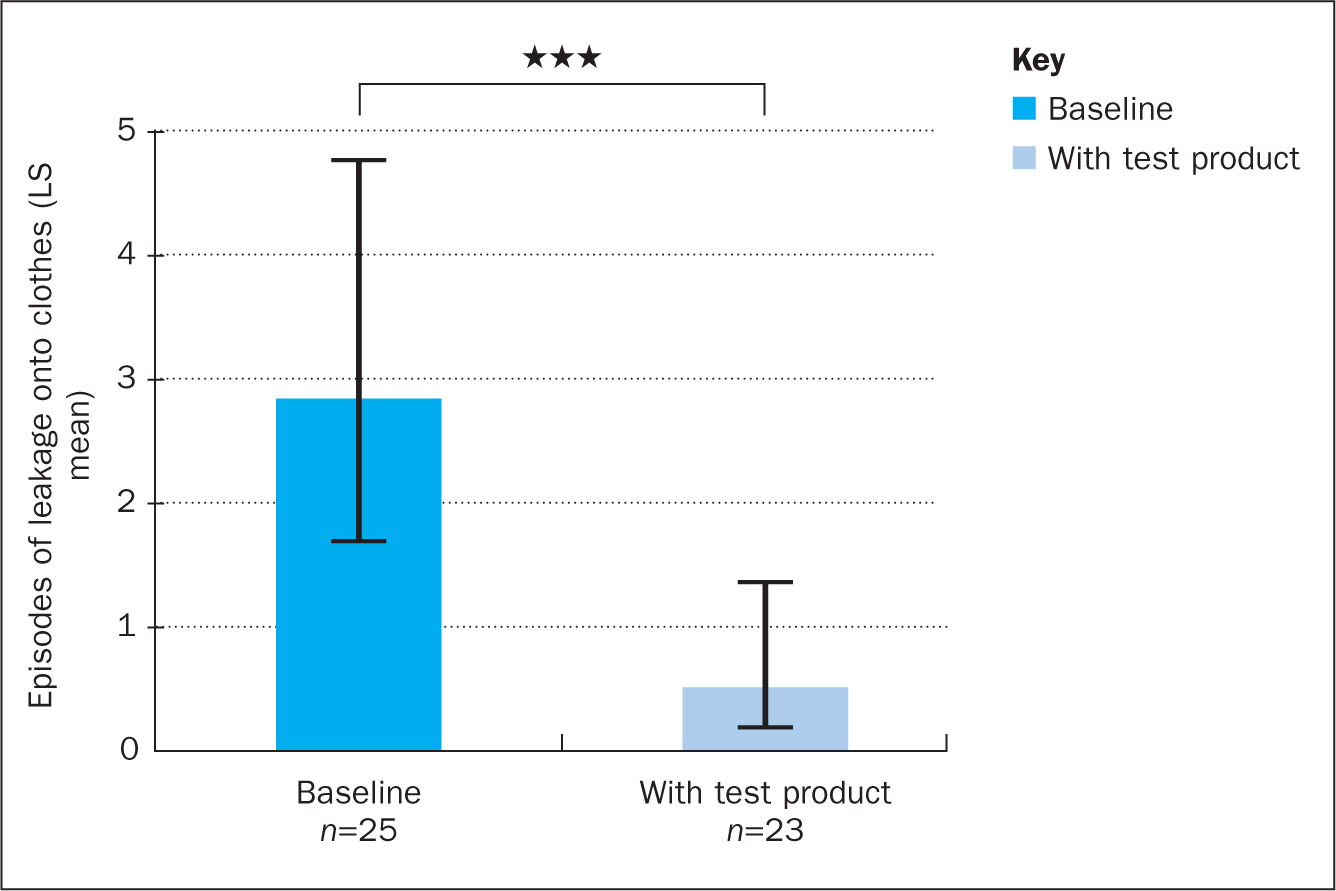
At baseline, subjects on average experienced 2.8 (range 0–15; 95% CI (1.7;4.8)) episodes of leakage onto clothes during the preceding 21 days. During the test period of 21 days the subjects experienced on average 0.5 (range 0-5; 95% CI (0.2;1.4)) episodes of leakage onto clothes, corresponding to an 82% reduction in episodes of leakage onto clothes (P<0.001) (Figure 5).
Feeling of leakage control
Most subjects (n=18, 78%) felt that the test product provided a higher feeling of leakage control compared with their usual product, and 70% (n=16) of subjects felt that the test product provided higher control of leakage progression (Table 2).
Table 2. Leakage control
| Questions | Higher (95% CI) | Lower | P value |
|---|---|---|---|
| Did test product give you a feeling of leakage control? | 78% (56%; 93%) | 22% | 0.011 |
| Did the test product give you a feeling of control of leakage progression? | 70% (47%; 87%) | 30% | 0.093 |
Users provided multiple assessments of the test product. Two options were available as answers to each of the questions, ‘higher’ than usual product and ‘lower’ than usual product. Looking at the 95% confidence interval, if the lower confidence limit is >50% it means that the test product was assessed positively by the majority of users (n=23)
Behavioural changes
Of the 360 baseplate changes recorded in the trial period, users stated that in 151 (42%) of the cases they had changed the baseplate due to a notification from the app, while in 196 (54%) of the cases the users followed their usual changing patterns.
Worry about leakage
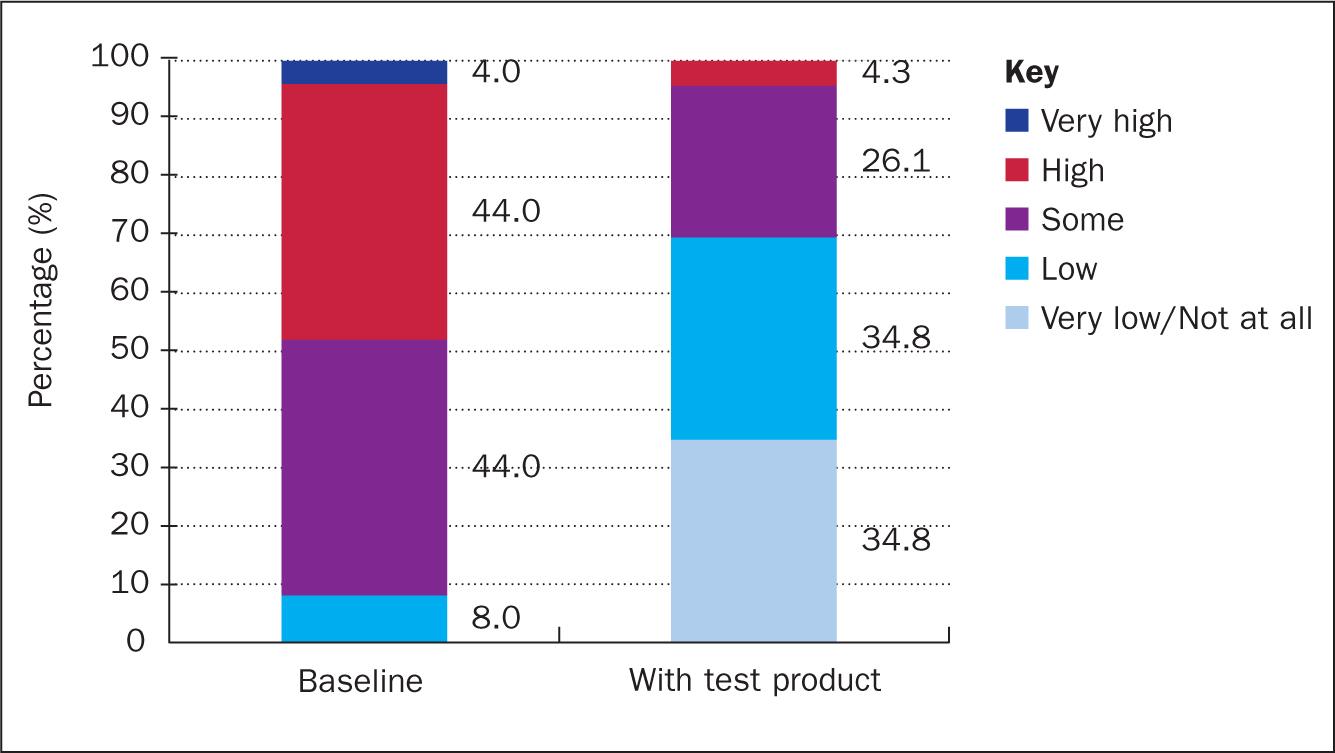
Half (48%) of the subjects worried about leakage to a ‘high’ (44%) or ‘very high’ (4%) degree at baseline (V1), whereas 44% worried about leakage to ‘some’ degree. The remaining two subjects (8%) worried about leakage to a ‘low’ degree (Figure 6). Subjects' level of worry about leakage decreased significantly from baseline (V1) to termination of the study (V2) (P<0.001). At termination of the study (V2), none of the evaluated subjects worried about leakage to a ‘very high’ degree and just over 4% worried to a ‘high’ degree, while 26% worried about leakage to ‘some’ degree. After the test period, most subjects (70%) worried to a ‘low’ or ‘very low’ degree (Figure 6).
Burden of leakage (OLI tool)
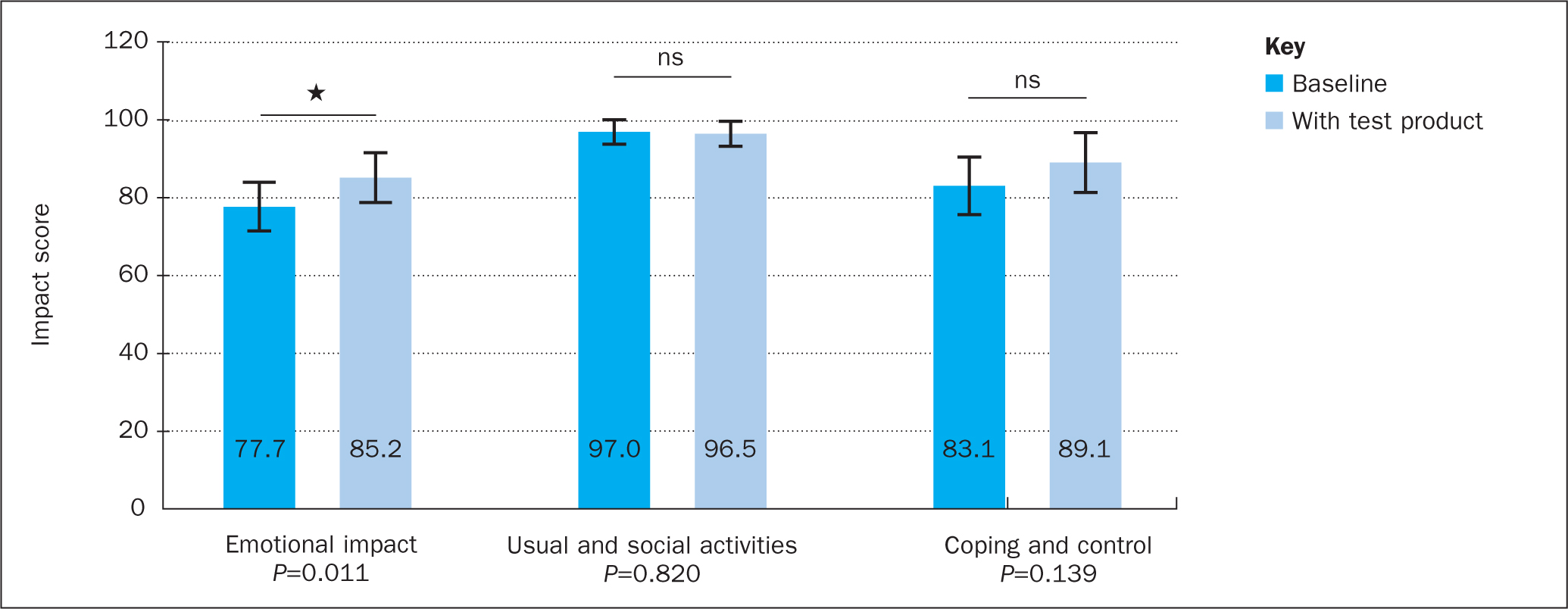
After 21 days of using the test product, subjects scored significantly better in the ‘emotional impact’ domain of the OLI tool (77.7 vs 85.2; P=0.011) compared with baseline levels. No significant changes in the ‘usual and social activities’ or ‘coping and control’ domains were observed (Figure 7).
Product-related quality of life (Ostomy-Q)
After 21 days of using the test product (V2), subjects scored significantly better in the ‘discreetness’ (P<0.001) and ‘confidence’ (P=0.033) domains compared with the evaluation at baseline (V1). No significant changes in the ‘comfort’ or ‘social life’ domains were observed. The combined product-related QoL score was significantly higher at V2 compared with V1 (Δ9.2; P=0.002) (Figure 8).

Sleep
The subjects were asked at completion of the study (V2) ‘did the test product affect your sleep?’. The proportion of subjects answering ‘Yes, for the better’ was 35% (95% CI (16%;57%)) (P=0.21). Most subjects (65%) (n=15) reported that the test product did not affect their sleep compared with their usual product. None of the subjects reported that the test product led to worse sleep compared with their usual product.
Safety
Two adverse events were reported during the study and were either considered ‘probably’ or ‘possibly’ related to the test product. Both adverse events were related to skin irritation, however they were both considered to be mild, and both resolved within 8–10 days. The adverse events did not affect the participants' continuation in the study.
Discussion
Despite continuous innovations in ostomy solutions, leakage of stomal effluent remains a major challenge in ostomy care. Digital health solutions, ie wearable devices and connected healthcare solutions, are increasingly being adopted in healthcare systems across many areas of patient care (Awad et al, 2021). Only a few digital health solutions have evaluated to improve ostomy care, including web-based patient support resources (Pittman et al, 2017), follow-up teleconsultations (Augestad et al, 2020), follow-up care via a mobile app (Wang et al, 2018), automated assessment of peristomal skin using artificial intelligence (Andersen et al, 2020) and sensor technology to measure effluent volume in connection with a remote monitoring platform (Kontovounisios et al, 2018; Fearn et al, 2020).
The present trial evaluated a novel leakage notification system enabling users to continuously monitor for leakage of effluent underneath the baseplate, by providing subjects with notifications of changes to their baseplate status via a smartphone app. During the 21-day trial period the subjects experienced an 82% reduction in episodes of leakage onto clothes compared with the estimated number of leakages onto clothes in the 21 days prior to the test period. Moreover, wearing the test product significantly decreased subjects' worry about leakage. The degree of worry about leakage has previously been correlated with the use of different accessory products, such as rings/seals, paste, tape and belts (Jeppesen et al, 2022). Thus, decreasing people's worry about leakage may potentially reduce the use of standard-of-care accessories that people with an ostomy might use to mitigate the risk of leakage, although this hypothesis needs further evaluation in future trials.
The observed improvement in leakage control and concomitant reduction in worry about leakage may have led to the observed improvements in QoL during the trial. Users experienced a significant improvement in the emotional domain (Δ7.5) of the validated OLI tool, which was equal to the minimal clinical important difference (MCID) (mean of three MCID values derived from different methods: Δ7.6), suggesting that the test product did indeed provide a meaningful change in the emotional domain for the subjects (Nafees et al, 2018). Due to the specific item questions included in this domain, it might be inferred that users experienced less stress, frustration and embarrassment with the test product compared with their usual product solution. In addition, subjects in this study gave higher scores in product discreetness and confidence domains of the validated Ostomy-Q after use of the test product. The overall improvement in product-related QoL (Δ9.2) was higher than the MCID (mean of three MCID values derived from different methods: Δ8.8) (Nafees et al, 2017), suggesting that the test product provided a clinically relevant change for the subjects.
A number of previous studies have also used the Ostomy-Q tool to assess QoL in people with an ostomy (Walker et al, 2016; Hedegaard et al, 2020). A retrospective survey with more than 3000 subjects showed that the higher the frequency of leakage underneath the baseplate, the lower the domain scores of the Ostomy-Q tool (Hedegaard et al, 2020). Moreover, a clinical trial investigating a convex ostomy appliance showed reductions in leakage frequency and leakage area, and concomitant improvements in QoL (Walker et al, 2016). Taken together these studies suggest an association between ostomy solutions that can reduce leakage incidents and an improvement in users' QoL.
Stoma care nurse consultations have previously been shown to help reduce the incidence of leakage (Porrett et al, 2011) and lead to improvements in QoL of people with an ostomy (Erwin-Toth et al, 2012). As highlighted in the present study, real-time status updates of possible leakages under the baseplate, enabled by use of a digital leakage notification system as a stand-alone solution, can reduce leakage episodes onto clothes and improve QoL. In future ostomy care, digital solutions, such as the current test product, may permit remote monitoring of patients' leakage data, and provide a complementary service to stoma care nurses. This may aid identification of users who struggle with frequent episodes of leakage and, once identified, nurses may assist them in re-evaluating their pouching system type, shape and use of accessories, to optimise fit and combat leakage.
Limitations
There were some limitations to the present study that may have impacted the results. The trial was a non-blinded, single-arm study in a small number of subjects already using products manufactured by Coloplast A/S, which may positively bias users' perception of new solutions developed by Coloplast A/S. Baseline values may be influenced by recall bias, since subjects were not told to monitor leakage frequency until engaged as actively participating in the study.
The study inclusion criteria limited entry to those people who worried about leakage to ‘some’ or higher degree and who were struggling with leakage. Therefore, subjects who do not struggle with leakage issues or worry about leakage to a lower degree may not necessarily benefit from this proposed solution to the same extent as reported in this trial. Nonetheless, two out of three people with an ostomy worry about leakage to ‘some’ or higher degree (Jeppesen et al, 2022), indicating that the study population in this case still represented a large proportion of the global population of people with ostomies. Future work will aim to evaluate the effect of the product on larger and more diverse populations.
Conclusion
This study shows that a novel digital leakage notification system can support users in their daily ostomy care by notifying them of a potential leakage and thus informing the decision of when to change the baseplate, and providing people with a feeling of leakage control. Across a wide demographic of users and various types of ostomy pouching systems, the tested system performed well and was found to be acceptable (4% drop-out rate). Performance accuracy was influenced by software bugs and user adherence to notifications. Software updates have subsequently addressed the bugs and accuracy is expected to increase accordingly. Use of the device resulted in fewer episodes of leakage onto clothes, reduced users' worry about leakage and led to improvements in QoL.
Although this pilot study provided positive indications, future larger studies with a longer test period and with users' own smartphones are required. Future studies should not focus solely on an experienced population or those with specific difficulties in leakage control, but also explore the impact of the system on self-management and stoma care routines in newly operated stoma patients and wider stoma patient populations. Whether there is translation of improved leakage control, through use of this novel digital leakage notification system, into potential economic benefits for the healthcare system, requires further research.
KEY POINTS
- For people living with an ostomy, leakage of stomal effluent is a key issue compromising stoma-related quality of life
- Use of a novel digital leakage notification system reduced the number of leakage incidents outside the baseplate
- Subjects reported reduced leakage worry and improvements in quality of life, reflected as less panic, less embarrassment, and an increased feeling of security
- Providing information regarding leakage seeping underneath the baseplate supported users in their ostomy care routines and in the timely change of baseplates
CPD reflective questions
- Have you tried to ask your patients how and to which degree are they limited in their daily life because of stoma effluent leakage or the worry about it?
- How often do your patients experience leakage incidents and what do they do to try to prevent these?
- Consider how many product changes and supporting products are used on a weekly basis to lower the worry of a leakage?


