In the past two decades, different types of cyanoacrylate glue (CG)—octyl-CG, butyl-CG and octyl-butyl-CG—have been used in surgical practice to close skin incisions, to repair lacerations of internal organs, to stop oesophageal bleeding and to secure prostheses for inguinal hernias (Singer and Thode, 2004).
Since 2000, octyl-CG has been widely used in the authors' university hospital for closing skin incisions after minor surgical procedures as an alternative to sutures and staples, with several advantages in terms of control of local bleeding, to provide a barrier to bacteria and for cosmetic results (Gurnaney et al, 2011; Auyong et al, 2017; Chalacheewa et al, 2021).
In 2007, Wilkinson et al reported for the first time the use of CG to stabilise centrally inserted central venous catheters (CICCs) as an alternative to sutures. The same authors reported the effectiveness of CG in reducing dislodgment of epidural catheters (Wilkinson et al 2007; Wilkinson and Fitz-Henry, 2008).
The benefits of CG include not only the sutureless stabilisation of the catheter but also a potential antibacterial activity, as proven in vitro—in particular for octyl-CG—especially against Gram-positive bacteria (Bhende et al, 2002; Narang et al, 2003; Wilkinson et al, 2008).
In the authors' 1300-bed university hospital, most central lines are inserted by a multiprofessional, multidisciplinary vascular access team. The authors started to use CG 10 years ago, mainly as a haemostatic strategy in patients at a high risk of local bleeding.
In 2012, they reported for the first time the effectiveness of CG in reducing bleeding from exit sites within the first 24 hours of peripherally inserted central catheter (PICC) placement. They studied 45 adult patients requiring the insertion of polyurethane PICCs without reverse tapering; no significant local bleeding at 1 hour and after 24 hours was recorded. Both octyl-butyl-CG and butyl-CG were used, and no local reactions were reported (Pittiruti et al, 2012).
The efficacy of CG in reducing the risk of bleeding was further studied in a group of adult patients requiring polyurethane CICCs and PICCs without reverse tapering, in a non-intensive care ward in the authors' hospital. The exit site of 65 central catheters (45 PICCs, 11 dialysis catheters and nine CICCs) was assessed at 1 hour and 24 hours after insertion. No local bleeding occurred. No local adverse reaction or damage to the polyurethane was reported (Scoppettuolo et al, 2013).
In the same year, the authors also carried out a randomised clinical study to evaluate the efficacy of two methods of sealing exit sites—metallic powder (Statseal, Biolife) versus octyl-butyl-CG—in patients in the intensive care unit. Thirty polyurethane PICCs without reverse tapering with double (5 Fr) and triple (6 Fr) lumen were randomised to be sealed with either metallic powder or with CG.
Bleeding was assessed at 1 hour and 24 hours after placement and weekly thereafter. All catheters were removed after 3 weeks, and cultures of both the subcutaneous tract of the catheter and the catheter tip were performed. There was no early bleeding at 1 hour, but there were two later bleedings (one in each group) at 24 hours. All cultures of the subcutaneous tract were negative. One PICC in the metallic powder group had a tip culture test positive for Candida albicans, although the blood culture was negative.
The authors concluded that both methods were effective in reducing bleeding at the exit site, and that the compliance of nurses and patients was higher for the glue. Preliminary data about the cultures suggested that sealing the exit site (with either method) was effective in reducing extraluminal contamination (Annetta et al, 2013).
Two experimental in vitro studies further confirmed the antimicrobial activity of CG. In 2012, the in vitro efficacy of octyl-CG and butyl-CG for the securement of intravenous catheters was studied (Simonova et al, 2012). While butyl-CG was associated with stronger securement than octyl-CG, both compounds showed significant antibacterial activity against Staphylococcus aureus and S. epidermidis. Another in-vitro study (Rushbrook et al, 2014) demonstrated the bactericidal property of octyl-CG against Gram-positive bacteria (but not against Escherichia coli, Pseudomonas aeruginosa or C. albicans).
In 2014-2015, three more clinical studies analysed the efficacy of CG in securing vascular access devices. Two randomised studies addressed the use of CG for peripheral arterial catheters. While the first study was inconclusive (Edwards et al, 2014), the second demonstrated that CG was associated with the lowest risk of catheter failure and was more cost-effective than other strategies, including bordered transparent dressings and/or sutureless securement devices (Reynolds et al, 2015). Similar results were reported in a randomised clinical study on short peripheral cannulas (SPCs) (Marsh et al, 2015).
Meanwhile, further clinical studies carried out at the authors' hospital demonstrated the high efficacy of both butyl-CG and butyl-octyl-CG in reducing bleeding at the exit site. In a prospective study on 45 PICCs without reverse tapering, the risk of bleeding was reduced from 40% to zero in the first hour and from 15% to zero at 24 hours after insertion (Scoppettuolo et al, 2015).
A wider retrospective study on 1429 central venous catheters was also conducted in the university hospital (Pittiruti et al, 2016). This study included: 348 non-tunnelled PICCs inserted in patients at a high risk of bleeding (those with chronic renal failure, hepatic dysfunction or coagulation disorders or receiving antithrombotic treatment); 165 non-tunnelled CICCs and femorally inserted central catheters (FICCs) in patients at high risk of local bleeding (including dialysis catheters); 114 tunnelled central catheters (PICCs, CICCs and FICCs, cuffed or non-cuffed); and 802 ports (both chest ports and PICC ports). At the end of the study, the authors concluded that CG was 100% effective in preventing post-insertion bleeding from the exit site in every type of central venous access device, even in patients at high risk of bleeding, in both children and adults. The use of glue for sealing the exit site of PICCs was found to be not only effective but also highly cost-effective, given that early bleeding would often mean an unscheduled dressing change was needed.
Considering the results of this retrospective study and the evidence accumulated in previous years (2012-2015), the university hospital's policies were modified. In 2016, the official hospital policies on the insertion and management of venous access devices recommended the use of cyanoacrylate glue for three main indications:
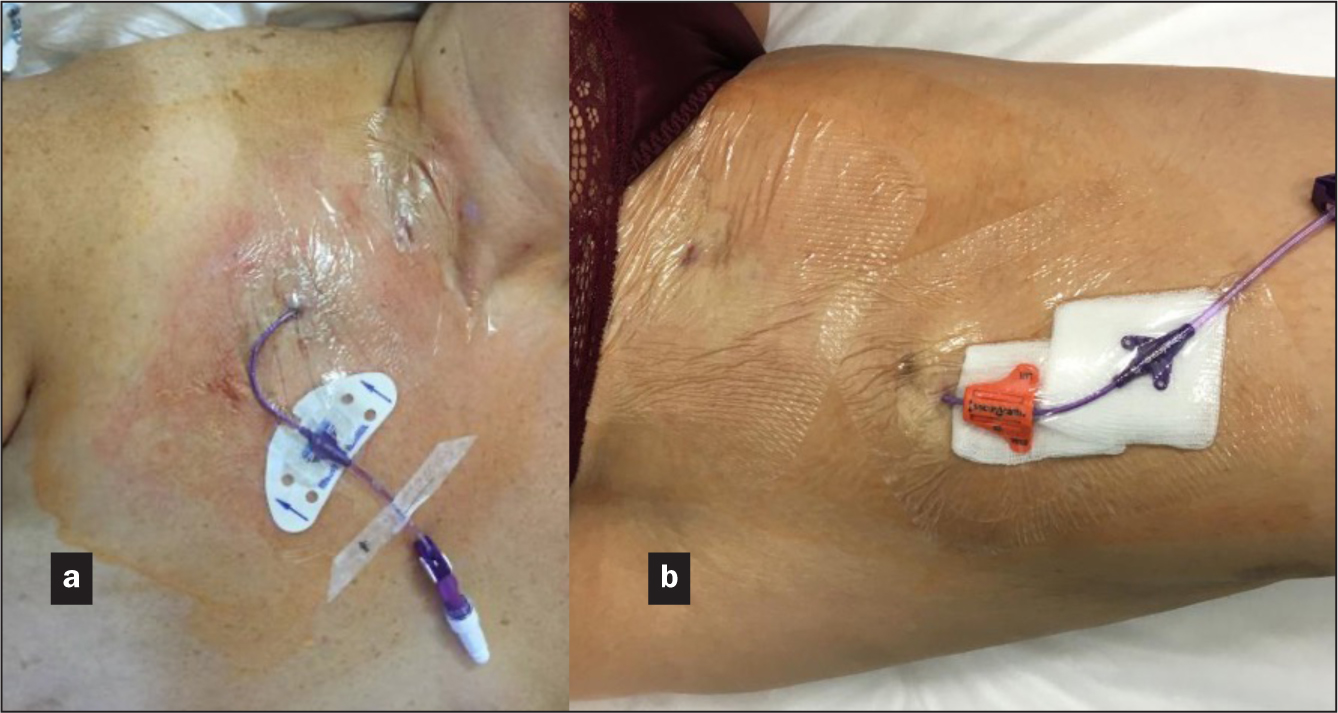
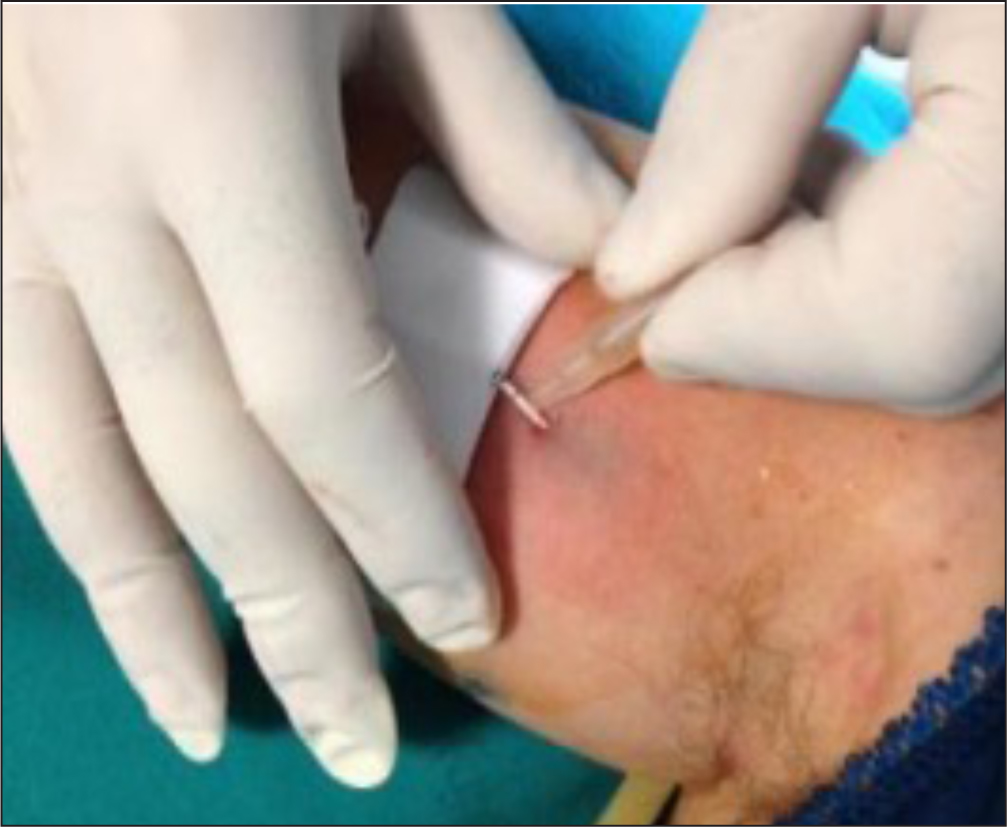
- Sealing the exit site of all PICCs soon after insertion (Figure 1)
- Sealing the exit site of other types of central venous access devices (CICCs, and FICCs) in patients at a high risk of local bleeding
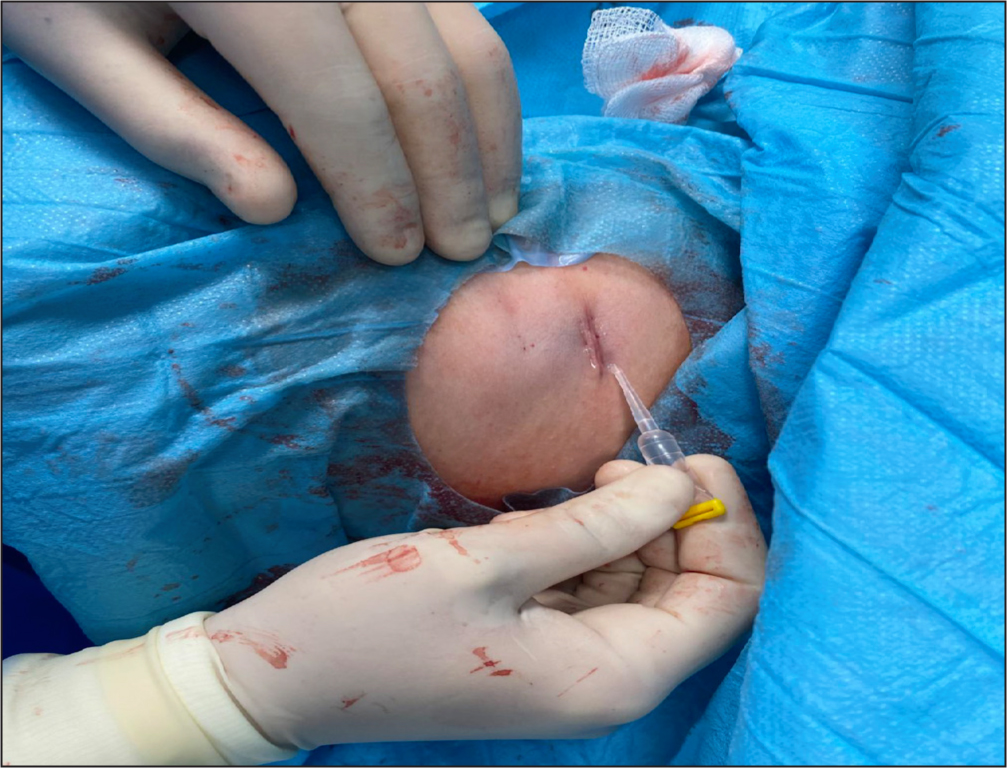
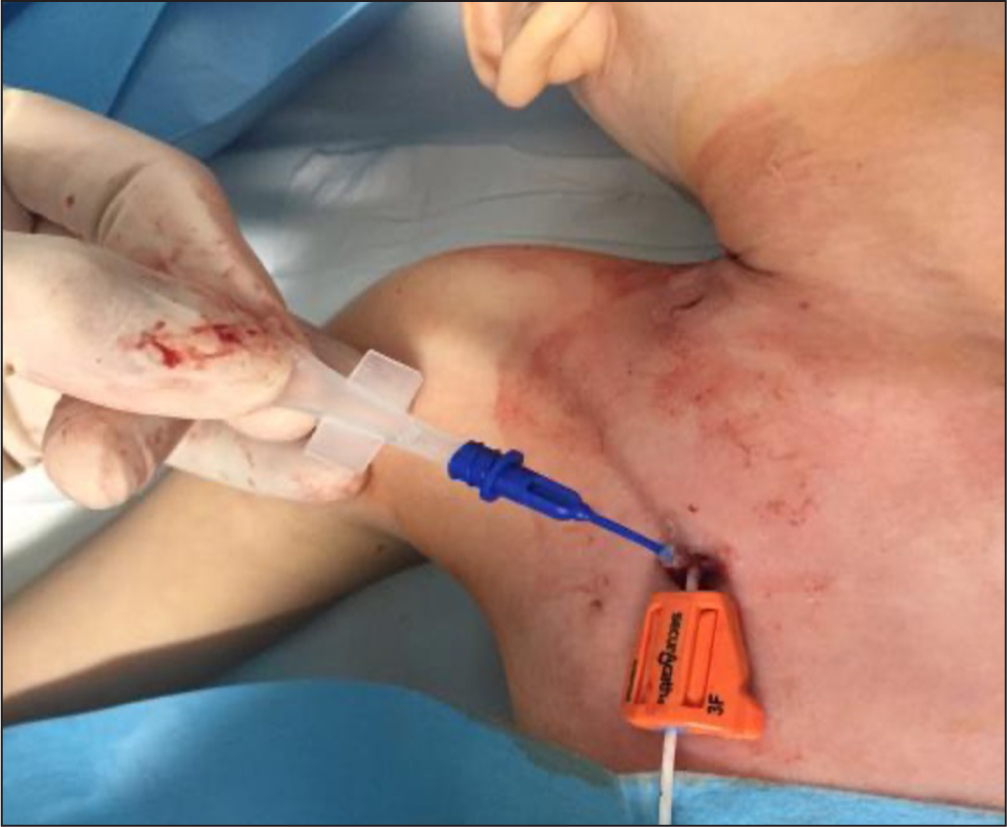
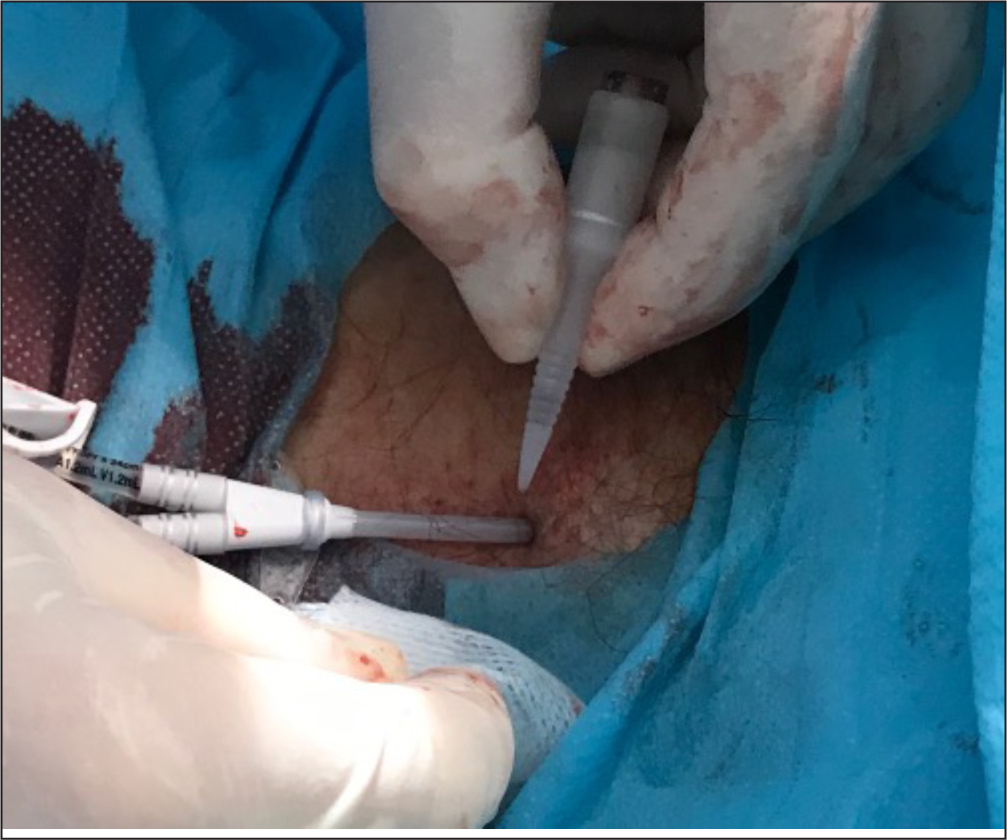
- Closing the skin incisions required for the implantation of tunnelled central catheters (Figure 2) and ports (Figure 3).
Expanding the indications: 2016-2019
After the 2016 hospital policies were introduced, the use of CG in venous access was implemented significantly in the hospital (2000 vials/year for an estimated 7000 central venous catheters inserted per year by the vascular access team).
Concerned that CG might potentially damage catheters, the authors carried out an in-vitro study analysing the chemical and physical interactions between octyl-butyl-CG and 12 brands of PICCs (11 made of polyurethane and one of silicone). The samples were analysed after 4, 8 and 12 weeks of contact with the glue. The conclusion was that the long-term use of octyl-butyl-CG was not associated with any damage to polyurethane catheters, although it may alter the physical properties of silicone (Di Puccio et al, 2018). Considering that use of silicone catheters had been abandoned at the hospital since 2010, the findings of this study were reassuring.
In the following years, further clinical studies focused on CG as securement device.
A randomised trial involving 380 SPCs inserted in 360 adult patients demonstrated that, when using CG, catheter failure was 10% lower and dislodgement was 7% lower than when standard dressings were used (Bugden et al, 2016). On the other hand, a randomised trial involving 221 cardiac surgical patients showed that CG with a standard transparent dressing was not effective for the securement of CICCs; however, when CG was added to standard care (sutures and polyurethane dressing), dislodgments were reduced from 4% to 0% in comparison with standard care only (Rickard et al, 2016). A single-centre study involving 124 PICCs in patients with cancer showed that securement by CG and standard transparent dressing was associated with the lowest risk of catheter failure (Chan et al, 2017). In a larger randomised trial involving 1807 patients that analysed different strategies of SPC securement in adults, the results were inconclusive, but some adverse events (skin damage) were reported in the CG group (Rickard et al, 2018).
At this time, other studies focused on paediatric patients. In a randomised trial involving 101 PICCs in children (Kleidon et al, 2017), CG was effective both in reducing catheter dislodgements and in decreasing local bleeding. A randomised trial comparing securement devices in 48 tunnelled central venous catheters in children (Ullman et al, 2017) was associated with inconclusive results. In another randomised trial involving 108 children with non-tunnelled central venous devices, securement by CG only was less effective than securement by sutures or sutureless devices (Ullman et al, 2019).
However, in a clinical study carried out in the university hospital's paediatric intensive care unit, an insertion bundle for avoiding extraluminal bacterial contamination of CICCs (cyanoacrylate glue with a sutureless device, tunnelling and a transparent dressing) was associated with a 90% reduction in catheter-related bloodstream infections from 15 to 1.5 episodes per 1000 catheter days (Biasucci et al, 2018).
This potential antimicrobial activity of CG was confirmed in several experimental studies (Bull et al, 2018; Prince et al, 2018; Waller et al, 2019). In one of these studies (Bull et al, 2018), the effectiveness of CG to inhibit bacterial growth at the extracorporeal membrane oxygenation (ECMO) cannulation site and in securing ECMO cannulas was evaluated in vitro. Bull et al (2018) concluded that combining CG at the cannula insertion site with a sutureless securement device could be an effective strategy to prevent or minimise both infection and line dislodgement.
The role of glue in venous access was discussed in a meeting organised by the authors' team in 2019, based on the literature and on an audit of their experience. They concluded that securement of central venous access devices by CG only was not effective and that, for CICCs, FICCs and PICCs, CG should be used not for securement but for the purpose of reducing local bleeding and bacterial contamination. For long-term central venous access, the combined use of CG, tunnelling and subcutaneous anchorage was judged to be especially beneficial because of a synergistic effect on the reduction of several complications (infection, dislodgment and bleeding) (Judge et al, 2018; Ortiz-Miluy et al, 2019).
The cost-effectiveness of CG (for both peripheral and central venous catheters) was related mainly to its haemostatic effect, with the elimination of a need for dressing changes 24 hours after insertion, leading to significant cost savings (Nicholson and Hill, 2019).
The authors also agreed that CG should be used in all central venous access devices requiring tunnelling (for closing the skin incision at the puncture site) or requiring subcutaneous placement of a reservoir (for closing the skin incision at the pocket) (Martin et al, 2017).
The present: 2020-2021
Over the past 2 years, many more clinical studies have focused on CG for both central and peripheral catheters and in different patient populations.
Regarding CICCs, a randomised clinical trial involving 121 adult patients (Mitchell et al, 2020) confirmed that CG was not optimal for the purpose of securement only. However, as another trial with 150 adult patients showed, when added to standard securement, CG may reduce the incidence of early dressing change (Prachanpanich et al, 2021).
Findings seem to be different for SPC. A large, randomised trial involving 350 adult patients (Bahl et al, 2021) suggested that a transparent dressing combined with CG may improve survival of an SPC if the line is expected to be used for more than 48 hours. In a randomised study with 330 children, both integrated securement dressings and CG were more effective than bordered transparent dressings in reducing catheter failure (Kleidon et al, 2020). Nonetheless, the greatest change in practice in the past 2 years has been the introduction of CG into neonatal intensive care units.
Butyl-CG and octyl-butyl-CG have proven to be safe and effective for the securement of epicutaneo-caval catheters (ECC) (Barone and Pittiruti, 2020; D'Andrea et al, 2021; van Rens, 2021). The local application of small quantities of CG seems to be harmless even in premature newborns, while it is highly effective in reducing dislodgement and preventing bleeding and oozing at the puncture site.
Data also suggest that CG may be associated with a reduction in the risk of central-line bloodstream infection in neonates, probably by reducing bacterial contamination via the extraluminal route. One of the authors' protocols on the use of CG for umbilical venous catheters (UVCs) in pre-term neonates is being investigated. Preliminary results suggest that the addition of CG to a standard UVC securement may reduce catheter dislodgment and tip migration (Pittiruti et al, 2020).
Finally, the antibacterial activity of CG has been further confirmed by two recent studies. In one clinical study on 102 PICCs, local application of CG at the exit site soon after insertion was compared with the local application of a chlorhexidine-releasing sponge dressing. Both strategies were effective in controlling bacterial colonisation; however, CG was more effective in reducing local bleeding and more cost-effective than the sponge dressing in the first week after PICC insertion (Gilardi et al, 2021).
In an experimental study, the antimicrobial properties of two formulations of octyl-butyl-CG were compared in terms of bacterial inhibition at peripheral ECMO cannula insertion sites: both were capable of inhibiting bacterial growth and migration of S. epidermidis (Pearse et al, 2021).
As a result, in the past 2 years, several evidence-based documents have included specific recommendations about the use of CG in venous access.
In 2020, the Italian Association of Pediatric Hematology and Oncology guidelines recommended the use of CG at the exit site of all central venous catheters in children. The rationale for this recommendation is that glue is safe, allows rapid and complete haemostasis at the exit site, reduces the incidence of dressing change and decreases micromovements of the catheter, so it may be useful in preventing thrombosis and infection. The use of glue alone may not reduce the risk of catheter dislodgment but, when used in with other securement devices, it may improve the dwell time of central devices (Cellini et al, 2020).
The 2021 Standards of the Infusion Nursing Society include CG as an option for securing SPCs, combined with a standard transparent dressing or an integrated securement dressing, in both adult and paediatric patients (Gorski et al, 2021).
Finally, the Consensus on European Recommendations on the Proper Indication and Use of Peripheral venous access devices stipulate securing ‘integrated’ SPCs and long peripheral catheters (LPCs) using either a bordered transparent dressing with an integrated securement system or a standard bordered transparent dressing after application of cyanoacrylate glue at the exit site. According to this consensus, local application of CG should also be considered in any patient at high risk of bleeding (Pittiruti et al, 2021).
Conclusion
In summary, there is now enough evidence on CG in several areas.
CG is an effective securement only for SPC (Kleidon et al, 2020; Bahl et al, 2021) and ECC (D'Andrea et al, 2021; Van Rens et al, 2021); it should be used not alone but in combination with a transparent semipermeable membrane.
CG is a safe, effective and cost-effective strategy for avoiding the risk of early bleeding and oozing after the insertion of any peripheral or central venous catheter (Pittiruti et al, 2012; 2016; Scoppettuolo et al, 2013; Guido et al, 2020).
In all central venous access devices, CG may reduce the risk of infection by decreasing bacterial contamination via the extraluminal route (Annetta et al, 2013; Biasucci et al, 2018, Bull et al, 2018; Gilardi et al, 2021), but its antibacterial use should be limited to the first week (Corley et al, 2017; Gilardi, et al 2021), since there is not enough evidence to show that weekly replacement of glue is harmless.
CG is an effective and cost-effective replacement for sutures when a skin incision needs to be closed (ie in tunnelled venous access devices and in ports)(Pittiruti et al, 2016; Martin et al, 2017).
Based on such evidence, in 2021 the Italian Group of Long-Term Venous Access Devices (GAVeCeLT) released specific recommendations for the use of CG (Table 1)(Pittiruti and Scoppettuolo, 2021).
Table 1. Italian Group of Long-Term Venous Access Devices recommendations on the use of glue in venous access
| 1. Use cyanoacrylate glue |
|
| 2. Remember to prefer butyl-cyanoacrylate or octyl-butyl-cyanoacrylate |
| 3. Use a minimal amount of cyanoacrylate glue and only at the time of insertion |
In spring 2021, a revised version of the university hospital policies was issued. This document recommends the use of butyl-CG or octyl-butyl-CG for venous access, while octyl-CG is considered appropriate for skin closure after minor surgical procedures only. Its recommendations include:
- Consider using CG for sealing the exit site of SPCs in neonates, in children and in any patient at high risk of local bleeding, before covering with a semipermeable transparent membrane (Figure 4)
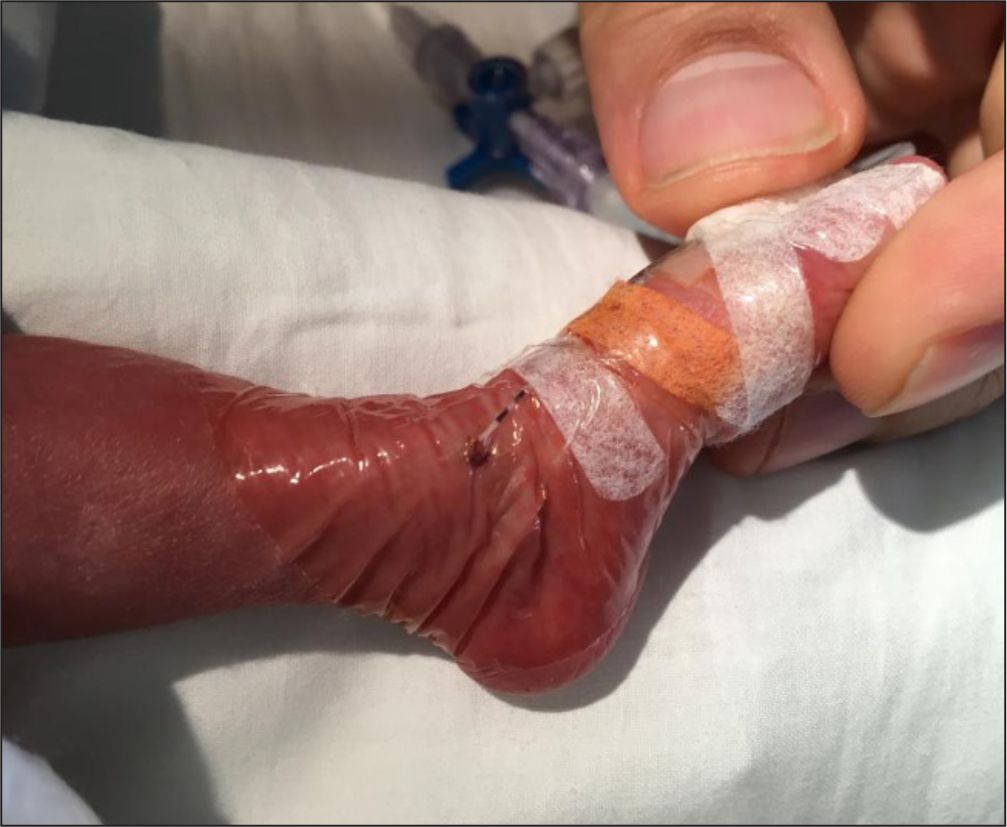
- Use CG for sealing the exit site of all ECCs in neonates, before covering with a semipermeable transparent membrane (Figure 5)
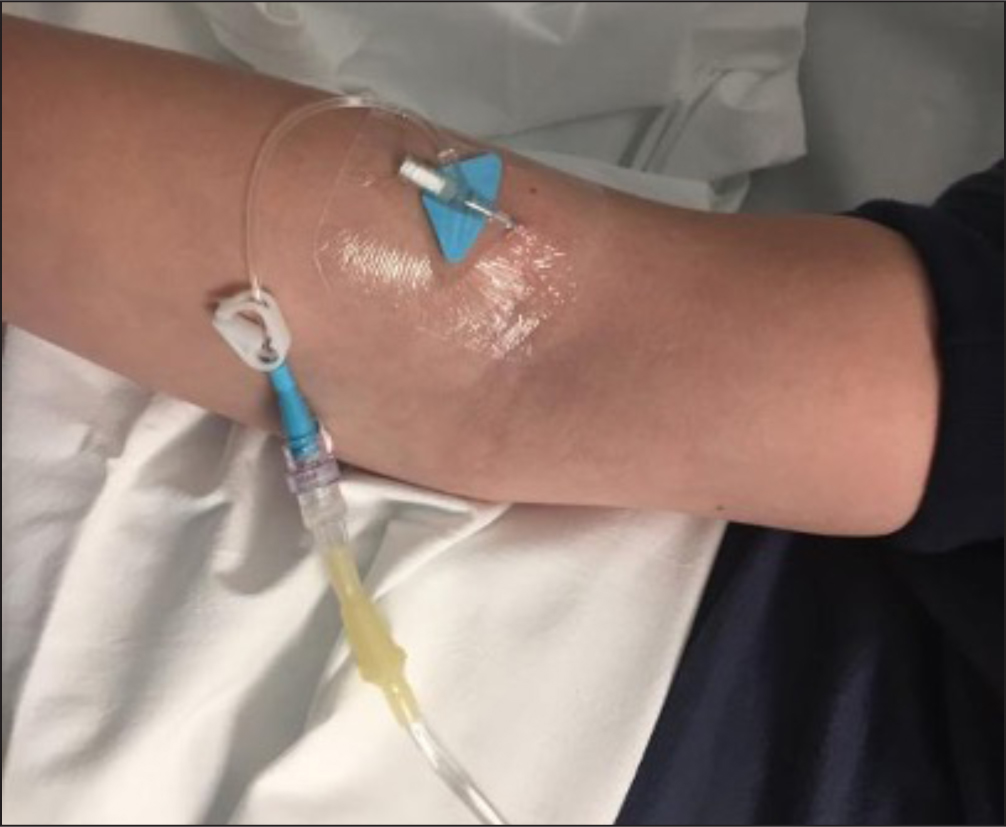
- Always use CG for sealing the exit site of any LPC, any midline catheter (Figure 6), and any central venous catheter (PICC, FICC or CICC), in both children (Figure 7) and adults (Figure 8), in addition to standard sutureless securement and coverage with a transparent semipermeable membrane
- Always use CG for closing any skin incision related to venous access procedures (e.g skin incisions required by tunnelled catheters and skin incisions for reservoir placement during port insertions).
In the authors' hospital, the vascular access team is currently using approximately 10 000 vials per year of butyl-CG or octyl-butyl-CG; no adverse effects related to CG have been reported in the last decade.
Prospective extensions of the use of glue (still to be supported by evidence) include UVC, peripheral arterial catheters and any SPC, as well as in patients at a low risk of bleeding/dislodgment. In addition, the safety and cost-effectiveness of weekly replacement of glue over the exit site of central venous access devices needs to be evaluated by future studies.
KEY POINTS
- Glue is effective for securement only for short peripheral cannulas and epicutaneo-cava catheters and when used with a transparent semipermeable membrane
- At all central venous access devices, glue is effective in minimising local bleeding and is cost-effective as it reduces unscheduled dressing changes
- Glue is probably effective in minimising bacterial contamination for all central venous access devices but should be used only in the first week as replacing it every week may be harmful
- To close skin incisions, glue is effective and cost-effective for all venous access procedures
CPD reflective questions
- Is cyanoacrylate glue (CG) completely safe on the skin at any age?
- What evidence is there that CG actually reduces local bleeding at the exit site?
- Is there evidence that CG may reduce bacterial contamination of the exit site?
- Which venous access devices may benefit from CG being used as a securement strategy?


