There are an estimated 1.2 million individuals living with chronic obstructive pulmonary disease (COPD) across England (Rayner et al, 2017). This is a significant increase from the 835 000 estimated in 2011 (Department of Health, 2011) and indicates that this estimate will continue to grow over the coming years. The northern regions of England, in particularly the North East and North West, have the highest and second highest prevalence, respectively (NHS Digital, 2017; Office for National Statistics, 2018). These regions contribute to making COPD the second highest cause of respiratory disease-related death in the UK, translating into the third highest rate of mortality across Europe (Snell et al, 2016; Marshall et al, 2022).
COPD is normally characterised by two main types: chronic bronchitis, which is consistent with a long-term cough with mucus, and emphysema, where the alveoli are weakened and tend to become damaged over time. People living with COPD experience symptoms that can regularly be reflected in a cough which is initially transient and unproductive in nature, but can become persistent over a longer period and can progress to a productive cough with frequent infections (Pauwels and Rabe, 2004; Nielsen et al, 2015). Individuals living with undiagnosed COPD will experience dyspnoea, which progressively worsens and often results in them visiting a GP, who will initially use spirometry measurements to diagnose the condition (National Institute for Health and Care Excellence (NICE), 2019). As dyspnoea worsens, it causes an adverse effect on people's activities of daily living and, as the disease progresses, people can often experience what they describe as a heavy weight when trying to breathe, a lack of air or a puffing feeling (Simon et al, 1990; Booth and Johnson, 2019).
The effects of living with COPD are well documented and as well as a cough and difficulty breathing, reduced mobility and poor appetite can be presenting problems as the condition worsens (Nielsen et al, 2015; Kwan et al, 2018). As COPD progresses and advances towards the later stages of the disease trajectory, weight loss and cachexia can occur from a higher-than-normal metabolic demand. This is because the body is attempting to increase its respiratory drive to oxygenate major organs and maintain homeostasis (Vassilakopoulos et al, 2004; Wüst and Degens, 2007; von Haehling et al, 2016; Kwan et al, 2018). von Haehling et al (2016) found that, across Europe and based on a combined population of 742 million people, an estimated 3.5% of the population were living with COPD, of which 1.4 million people were described as experiencing cachexia.
Cachexia related to COPD has been seen to increase mortality risk by up to 25%. Alahmad and Gibson (2023) examined data from 1.4 million COPD hospital admissions and determined the cachexia prevalence was 7.9%, affecting older patients but with no differences between genders. Hospital-associated complications, including cardiac arrest, were seen in those displaying signs of cachexia and, as a result, an increase in length of stay. Cachexia can occur in people with COPD who are frequently breathless. People can struggle to eat and drink, feeling like this activity is further exacerbating the feeling of not being able to breathe. This is because during the normal process of eating and drinking, the airway is closed and protected to avoid aspiration of food and fluid entering the lungs. This restriction on the airway can causes distress for some people living with COPD (Lancaster, 2015; Nagami et al, 2017).
Malnutrition
Current figures relating to malnutrition in the UK reveal that up to 3 million people at any one time are living with malnutrition (British Nutrition Foundation, 2023) and 93% of malnutrition occurs in the community (Holdoway et al, 2021). A third of people aged 65 and over who are admitted to hospital are at risk of malnutrition, and 50% of people from care homes admitted to hospital are also at risk. It is therefore imperative to understand the community context to malnutrition.
Murphy et al (2018) recognised the impact of an ageing population and identified that, with ageing and disease-related conditions, such as a reduction in physical activity/ability and psychosocial decline, come challenges in maintaining a good nutritional status. In a cross-cultural study of frailty between participants in Spain and the UK, Kelaiditi et al (2020) found a correlation between frailty and malnutrition in the community and demonstrated an overall prevalence of malnutrition between 19% and 23%. Frailty was more prevalent in the UK and associated with low BMI and financial income. Norman et al (2021), in a review of malnutrition in older adults, recognised the significance of both acute and chronic diseases, such as cancers, respiratory diseases such as COPD and circulatory disorders, and its relationship with impaired nutritional status. In 2006, NICE published its first guideline relating to oral, enteral, and parenteral nutrition, which was updated in 2017. The national guideline recognises the causes of malnutrition and its relationship with illness and impact on wellbeing (NICE, 2017).
Consequences of poor nutrition
Although Norman et al (2021) recognised the important impact of chronic conditions such as respiratory disorders have on the nutritional status of those living with the disease, Nguyen et al (2019) specifically reviewed the correlation between COPD and malnutrition. In a study of 168 outpatients living with COPD, they found that 74% were displaying signs of malnutrition, which was strongly associated with the progression of the disease. The prevalence of malnutrition for people living with COPD in an outpatient setting has been reported at 10-45% (Collins et al, 2013). However, Deng et al's (2023) study found that the percentage of people living with COPD with malnutrition or at risk of malnutrition ranged from 30% to 50%.
The effect of malnutrition on a person living with COPD can cause frequent exacerbations of the condition, hospitalisation, and a worsening of the person's physiological condition (Collins et al, 2018). Stratton et al (2003) recognised that weight loss in people living with the disease is common because of the increased energy expenditure needed to try to maintain resting respiratory function and the impact of the difficulty breathing presents to adequate dietary intake. This often worsens with age and disease progression and can present in a multifactorial physiological impact on the body and undoubtedly influences the overall physical and psychological wellbeing of the person living with the disease (Figure 1).
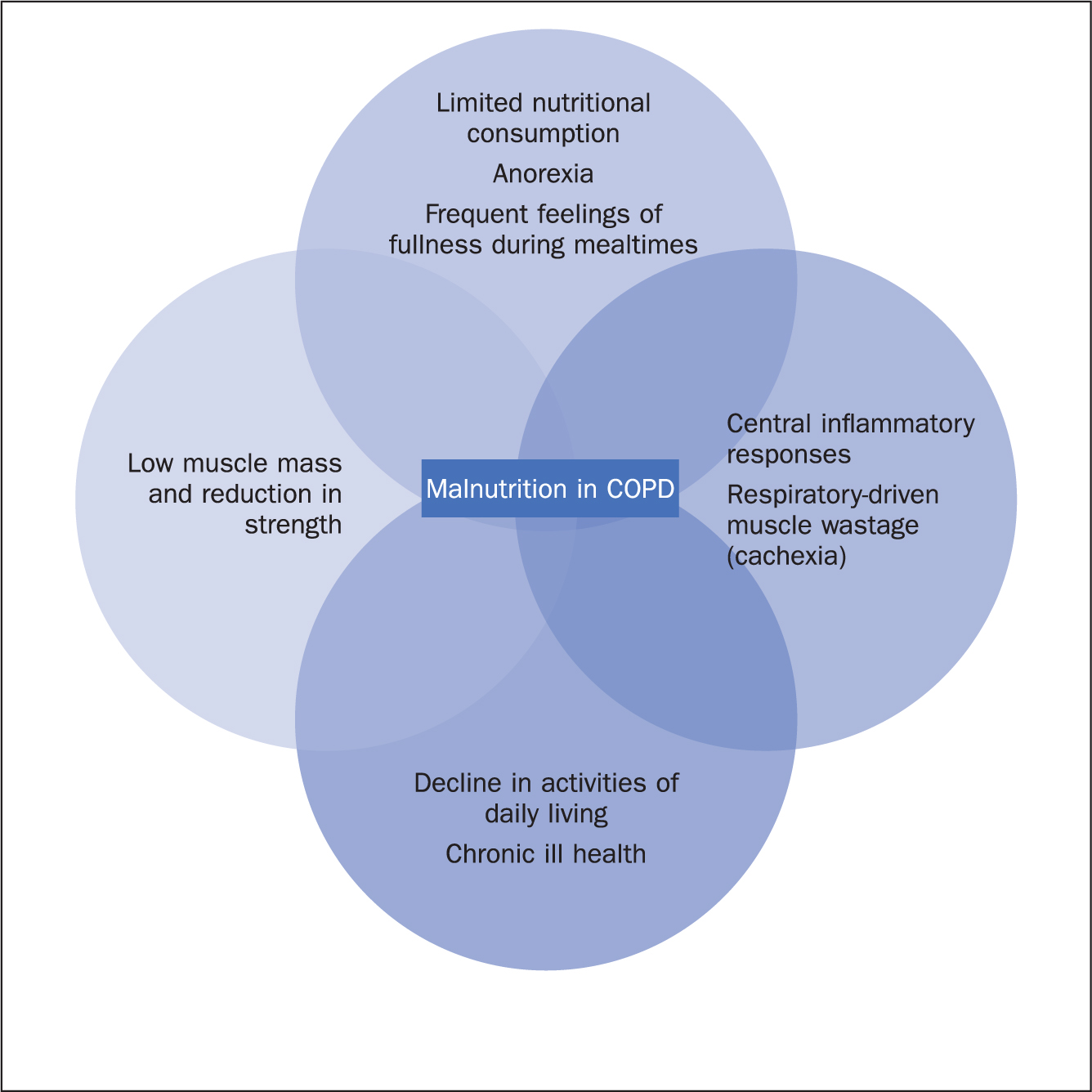
This connection between malnutrition and COPD has been found to affect the overall quality of life of those living with the disease – the increased risk of fatigue, apathy, increased risk of falls, social isolation and depression being just some of the consequences. Poor nutrition has ultimately been shown to be a predictor of increased mortality in this group of people (Perrot et al, 2020; Dávalos-Yerovi et al, 2021).
COPD care in the community
The care of people living with COPD predominantly takes place in the community under the care of general practices (Stone et al, 2022). Although the four nations of the UK have devolved healthcare policies, the principles for the diagnosis and initial management and treatment within primary care are directed by an overarching global guideline – the Global Initiative for Chronic Obstructive Pulmonary Disease (GOLD) (2023). Within England, the NICE Clinical Guideline NG115 and the Quality Outcomes Framework (QOF) for general practice drives the delivery of care, quality and income generation within this sector (NICE, 2019; 2023; Bostock, 2023). The QOF provides financial incentives for what it describes as quality care indicators being delivered within general practice. The measures and interventions for those living with COPD range from a clinical diagnosis, careful history taking, spirometry, assessing dyspnoea using a scale (Bestall et al, 2019), vaccinations and regular ongoing reviews recorded (Rodman, 2021; NICE 2023).
Managing long-term conditions in general practice is a fundamental role of the general practice nurse (GPN) GPNs are crucial in co-ordinating and managing many long-term conditions that have been moved from hospital-based care to primary care in recent years (Fanning and Berry, 2016). Chronic conditions including that of COPD were handed over to general practice with the Queen's Nursing Institute (Fannin and Berry, 2016) recognising that GPNs were providing a key role in caring for those living with COPD, including the diagnosis, treatment and ongoing management and follow-up care. Zwar et al (2016) recognised the important role GPNs play in the care of people living with COPD. In a study of 55 GPs and 55 GPNs, they found that nurses would assess, treat and prescribe as well as develop robust management plans for this group of patients, often independently of the GP. However, much of the research within this field predominately focuses on the biomedical elements of care, such as undertaking spirometry, forced expiratory volume in 1 second (FEV1), blood biochemistry and recording muscle strength, all components of the QOF (NHS England, 2023).
Gardener et al (2018) believed there was a need to explore more than a just a biomedical model of caring for a person living with COPD. They reviewed the literature relating to the support needs of those living with COPD, and recognised living with the disease requires a more holistic view and interventions that are as equally as important as those prescribed in health policy (NICE, 2019; 2023). As far back as 1998, Cadman and Findlay (1998) specifically recognised the lack of confidence in GPNs in relation to providing dietary advice to patients in general practice. In a study of Swedish primary care nurses, Odencrants et al (2007) acknowledged the fundamental relationship that nutrition has with COPD. They found that primary care nurses do not focus on nutrition as part of the care of COPD patients, often because of a lack of knowledge and confidence relating to this aspect of care.
This finding is reflected further in medical education, with little time given to disease-related malnutrition content, which in turn results in a lack of knowledge and confidence surrounding nutritional care (Broad and Wallace, 2018; Macaninch et al, 2020). Zwar et al (2016) noted that, although GPNs play an integral part in the care and management of individuals living with COPD, nutrition, despite being a fundamental nursing role, is often overlooked and considered secondary to the biomedical needs of the individual, with a focus on FEV1, blood biochemistry, and muscle strength.
This approach, paired with the historical understanding of COPD's late-stage disease progression and weight loss, has led to the view that malnourishment is a fundamental part of COPD in the late stages (Rimington, 2019). This view is reinforced by the QOF and the funding provided to GP practices, where nutrition in COPD remains unlisted as a key metric, despite clinical pathways and guidance detailing the need to consider and actively target malnutrition in individuals living with COPD (NICE 2019; 2023; Anderson et al, 2020).
Assessment of nutritional status
An assessment of nutritional status is recommended in the NICE (2017) guideline and promoted by BAPEN (British Association of Parenteral and Enteral Nutrition). BAPEN developed the Malnutrition Universal Screening Tool (MUST) as the first step in identifying someone with actual or a potential risk of malnutrition (BAPEN, 2021). The concept of nutritional screening, and the MUST tool in particular, is recognised to support a holistic approach to assessing a person who may require nutritional interventions. It aims to help direct care in the form of a management plan (House and Gwaltney, 2022). The nutritional tool (MUST) recommended by NICE (2017) comprises five steps (Box 1):
Box 1.The Malnutrition Universal Screening Tool (MUST)
| Step 1 | Measure BMI score |
| Step 2 | Calculate percentage weight loss score |
| Step 3 | Calculate acute disease effect score |
| Step 4 | Determine overall risk of malnutrition |
| Step 5 | Follow management guidelines |
Source: adapted from NICE (2006) and BAPEN (2021)
MUST consists of three key measures: the patient's body mass index, percentage weight loss within the last 3-6 months, and the acute disease effect score. The purpose of MUST is to combine the scores from these measures to provide a sensitive approach regardless of the assessor, in helping practitioners to identify individuals who could be displaying signs of malnutrition or highlighting those at risk of developing malnutrition. Much of the initial work undertaken to develop the tool was in hospital inpatient and outpatient settings (Stratton et al, 2003). Since then, further work has been undertaken by researchers to apply the MUST in various community settings, including residential care homes and those in sheltered housing (Harris et al, 2008; Mountford et al, 2016), which helped to provide management plans for people once screening and identification takes place. It is therefore reasonable that NICE (2017), recommends MUST as the tool of choice in screening for malnutrition in its guidance.
Evidence and management of nutrition in COPD
As highlighted, malnutrition for people living with COPD is a major issue and one that affects significant numbers of people with the disease (Deng et al, 2023). Despite the lack of a nutritional QOF indicator within current quality standards for general practice (NHS England, 2023), there is evidence that screening, assessment and planning can all positively influence those living with this chronic degenerative condition. The Malnutrition Pathway, developed specifically for COPD (Anderson et al, 2020), highlights three fundamental principles in the management of nutrition for those living with disease (Table 1). The pathway was developed by an expert working group of professionals including dietitians, nurses and medical staff and is endorsed by specialist respiratory groups, the British Lung Foundation and, more importantly for primary care, by the Royal College of General Practitioners. The task in hand is to facilitate the awareness of the pathway and to empower those in general practice, particularly nurses, to feel confident to explore opportunities to embed this in COPD patient reviews that already take place. The pathway provides a structured approach to managing malnutrition for those living with the disease and guidance on when to refer to more specialist support.
Table 1. Three key principles of nutritional management in COPD
| Goal setting |
|
| Management of malnutrition | Follow the guidance (Anderson et al, 2020) and observe the different strategies dependent on malnutrition risk categoryAim to increase nutrient intake in dietary advice provided to COPD patients, providing oral nutritional supplements, textured modified diets, or assistance with eating where appropriate, to ensure that weight loss is minimised, and functional tests improve |
| Monitoring progress | Aim to monitor the progress of each patient, modifying the nutritional interventions as appropriate, monitor nutritionally as per the NICE Guideline CG32 (NICE, 2017). Take weight change, hand-grip strength, and ability to undertake activities of daily living into account, also consider aspects such as physical appearance, appetite, and disease progression where appropriate |
It has often been thought that poor nutritional status is inevitable in the mid to end stages of the disease and an inevitable part of the disease process. However, as Collins et al (2012) and Collins et al (2013) have demonstrated in their review of the literature, there are significant benefits to providing nutritional support for people living with stable COPD in an outpatient setting. The studies examined those living with COPD and receiving oral nutritional supplements (ONS) in the form of ready-made drinks or supplementary enteral tube feeding. Participants received an extra 14.8 g of protein and 236 kcal daily in addition to their normal diet. The studies demonstrated improvements in nutritional intake, nutritional status, weight gain, lung function including capacity and strength, and quality of life.
In contrast, Ingadottir et al (2019) examined COPD patients in the hospital setting and 12 months after discharge. The small hospital feasibility study for those who had been identified as at risk, compared snacks versus oral nutritional supplements between meals. The results demonstrated that both groups gained weight and improvements were seen in quality of life. However, sustainability at 12 months was seen greatest in the snack group, possibly because snacks were easier to include as part of normal dietary habits than a prescription protein supplement. Bernardes et al's (2022) review of the evidence examining interventions of increasing energy and protein intake in COPD using food fortification and/or oral nutritional supplements found positive results. There was evidence of increases in weight, lean body mass, mid-arm circumference, triceps skinfold and handgrip strength, all measures used when undertaking an in-depth nutritional assessment on a person compared with control diets (Bernardes et al, 2022). Nutritional support for those living with COPD could offer an opportunity to improve not only the physiological function but also the quality of life of a person with the disease. It is important to ensure a thorough individualised assessment of nutritional status takes places, while recognising the stage of the disease process, to get maximum benefit of such interventions.
Conclusion
The prevalence of malnutrition among those living with COPD is significant and the impact this has on their lives affects physiological function, disease progression and quality of life. The assumption that the disease trajectory involves an acceptance of poor nutritional status is being challenged, with an increasing interest in research in this area. Nutritional screening and further assessment are effective ways of identifying those who may be displaying actual or potential risk of malnutrition. As people living with COPD are largely cared for in their communities by general practice, we need to recognise that quality indicators within this setting clearly drive practice in the care of this group. Patients living with COPD would benefit from a quality indicator that provides an incentive for the inclusion of nutritional care. Early treatment in the disease process may help to tackle some of the statistics we see relating to malnutrition for those living with COPD and challenge the negative assumptions.
KEY POINTS
- Malnutrition in chronic obstructive pulmonary disease (COPD) can cause frequent exacerbations and hospital admissions
- Patients living with COPD should be routinely assessed for actual and potential risk of malnutrition, including from the time of diagnosis, aiming for optimum nutritional status even when living with stable COPD
- Malnourishment should no longer be considered a standard part of late-stage disease progression in COPD and can be managed through assessment and screening
- The Malnutrition Pathway guidance should be used when implementing nutritional assessment and management in COPD
CPD reflective questions
- What do you currently do to incorporate nutritional care into your practice when caring for people living with COPD?
- Consider how you can promote nutritional assessment and management with your patients, how would you encourage increased nutritional intake and implement the guidance?
- How can you as a practitioner, promote nutritional management of patients living with COPD in the community despite it not being a funded marker on the Quality Outcomes Framework?
- What are the benefits of you providing nutritional assessment, support and management to patients living with COPD?


